Ben Valsler
This week, Kat Arney brings us a compound you’ll be all too familiar with if you have certain bad habits…
Kat Arney
Ever picked clotted blood off a cut or graze? If you’ve got a microscope handy, have a look and you’ll see that the clot is full of stringy fibres. This is an insoluble biological polymer called fibrin, made up of soluble subunits of fibrinogen. It’s a large sugary protein that normally circulates in the bloodstream – at a hefty two to four grams per litre of blood – until it’s activated and polymerised at the site of a wound by a complex cascade of molecular events.

As long ago as the fifth century BC, doctors noticed fibrin fibres floating in the blood – in fact the famous Greek doctor Hippocrates thought that blood clotting occurred due to cooling of the blood in contact with the outside world. The great 17th century Italian biologist Marcello Malpighi used an early microscope to zoom in on blood clots, noting that they consisted of a tangled web of fibres and trapped red blood cells.
It wasn’t until the turn of the 19th century that these strings were finally named ‘fibrin’, by French chemist Antoine Fourcroy, whose research led him to believe that animal matter contained three types of protein: albumin (mostly found in eggs, blood and milk), fibrin (in blood clots and muscles), and gelatin (in skin, tendons and similar tough stuff).
But only in 1847 did German doctor and polymath Rudolf Virchow finally coin the name fibrinogen, using the term to refer to molecules in the blood that appeared to react with oxygen in the air to form fibrin. French scientist Prosper-Sylvain Denis also has a claim to the discovery, as he independently suggested the named ‘fibrinogène’ in 1859, in his wonderfully-titled book Mémoire sur le sang, or memoire on the blood. It took another two decades for anyone to actually purify this mystery molecule, when Swedish biochemist Olof Hammarsten finally managed to extract it from horse plasma using a solution of regular table salt.
Over the next century, scientists began to dissect this useful molecule in great detail. They discovered that it’s made up of three separate protein chains, bound together to create a structure that’s a bit like a crab, with claw-like structures sticking out of each side, and some long ‘legs’ dangling down in the middle. When fibrinogen gets activated at the site of a cut, another protein called thrombin cuts the ‘legs’, revealing binding sites for the ‘claws’ on neighbouring molecules and starting the polymerisation process.
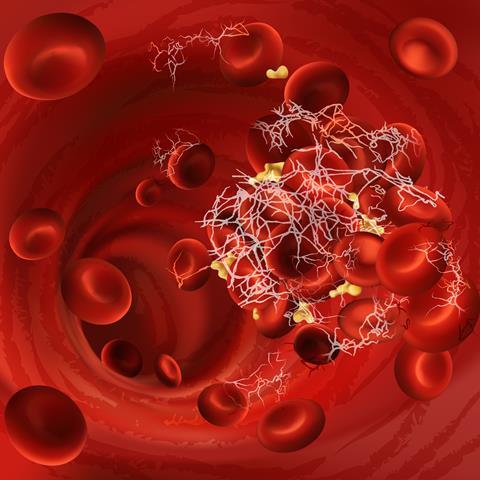
Careful chemical and genetic analysis has also revealed that clotting requires a complex cascade of proteins, originally named clotting factors I to XIII (and usually designated in Roman numerals). As befits such an important cornerstone of clotting, fibrinogen is Factor I. Defects in many of the genes encoding these factors result in haemophilia – a disease in which the blood fails to clot properly. Even the smallest wound can become serious, and it can be fatal.
Factor I, or fibrinogen, deficiency is a very rare but very serious genetic condition. Luckily, the treatment is relatively straightforward: infusions of concentrated fibrinogen purified from human blood plasma. This was first approved for use in Brazil in 1963, and is now used widely around the world with lifesaving results. It’s now pasteurised to reduce the chances of viral infection, and a staggering 3 million grams – that’s 3,000 kilos – have been used since the mid-1980s.
There’s also the converse problem – having too much fibrinogen in the blood. This is linked to an increased risk of coronary heart disease (as clots form in the arteries supplying blood to the heart) and also strokes, which are due to clots blocking crucial blood vessels in the brain.
There’s a final step in the story of fibrinogen, and its polymer fibrin, and that’s what happens when the blood clot has done its job and everything is healed up. Another protein called plasmin chops up the insoluble fibrin polymers to release small soluble proteins which get cleared out of the body. This usually happens within around 10 to 14 days - unless of course you’ve been naughty and been picking at your scabs.
Ben Valsler
Kat Arney on fibrinogen and its polymer, fibrin. For next week’s podcast, Katrina Krämer spoke to David Leigh at the university of Manchester about rotaxanes – unusual supramolecular structures that form the basis of some molecular machines…
David Leigh
Why can’t we have molecular robots to actually construct things for us? People are starting to move in that kind of direction.
Ben Valsler
Hear more from David Leigh as Katrina Krämer introduces the rotaxanes in next week’s podcast. Until then, get in touch with any suggestions – you can email chemistryworld@rsc.org or tweet @chemistryworld. And you can find more compounds to consume at chemistryworld.com/podcasts. Thanks for listening, I’m Ben Valsler.












No comments yet