While chemistry usually follows the downhill pull of thermodynamics, Alison Wendlandt is creating higher‑energy stereoisomers in the final stages of synthesis
Alison Wendlandt describes her work as sitting in the overlap of a Venn diagram. In one circle are stereochemical editing reactions that, for example, flip the handedness of a specific chiral centre. In the other are reactions that give products that are ‘contra-thermodynamic’, or higher in energy than the alternative stereoisomer. Her group coined the term ‘contra‑thermodynamic stereochemical editing’ in 2022 to capture this intersection of the two ideas, though she notes that a few similar methods existed before then.
‘The way that we view stereochemical editing is as a kind of complementary synthetic logic,’ Wendlandt says. Rather than building in 3D geometry while creating the molecular backbone itself, she develops tools that allow her to fine-tune stereochemistry in the later stages of synthesis. This can reduce the number of steps to synthesise certain stereoisomers or unlock previously unobtained configurations.

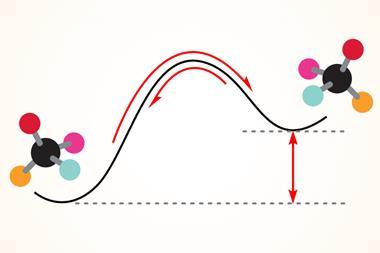
While there are many ways to do this, including using enzymes or mechanochemistry, Wendlandt predominantly opts for photocatalysis. Exposing photocatalysts to light creates long-lived excited states that allows electrons to move between a substrate and the catalyst, often creating substrates with, for example, weakened C–H bonds on chiral carbon atoms. This then means that the photocatalyst can abstract hydrogen atoms to create a radical intermediate. Tuning the catalyst and reaction conditions ensures that as the substrate decays back to the ground state, the hydrogen atom returns to the alternative face of the planar radical, leading to the opposite, higher energy enantiomer. This is one way to obtain contra-thermodynamic products, which can often be hard to synthesise with traditional stereoselective reactions.
‘We really are reaching a place in organic chemistry where we can make any molecule that we want, and now we’re sort of seeking these truly precise changes,’ says Wendlandt. She likens her work of fine-tuning organic molecules to base pair editing, a technique that allows scientists to directly swap a single DNA base pair for another, without needing to pull the two DNA strands apart.
Sugar tweaking
When Wendlandt started her independent research career, she says that she was keen to work with carbohydrates. To her, these molecules seemed like ‘the next frontier of biomolecules, where chemistry is really the limiting step in many ways’, including site- and stereoselectivity.
One of Wendlandt’s earlier studies showed how using an iridium-based photocatalyst, a thiol and a bicyclic amine could collectively invert the stereochemistry of specific chiral centres in unprotected sugars, oligosaccharides and glycans.1
The team was able to use this photocatalytic set-up to, for example, make d-allose from d-glucose in one step. This involves moving one alcohol group from an equatorial to an axial position, which increases the number of unfavourable steric interactions and raises the energy of the product. d-allose is a hard-to-synthesise sugar that can slow down the growth rate of certain solid tumours, such as glioblastomas.2 Alternative strategies to make this sugar include using existing methods to synthesise it over several steps or using low-yielding enzymes to do the conversion.
Wendlandt’s interconversion reached a yield of around 30% at a 1g scale. ‘I really try to sort of beat this yield mindset out of my students,’ says Wendlandt. ‘If you can make a thing that you couldn’t actually ever make before, or save 15 steps, then a 20% yield is awesome.’
However, as subtle tweaks can lead to products with very similar properties, Wendlandt explains that her group has had to come up with new ways to separate isomers. She recalls one student reacting amine–functionalised silica with boronic acid to create a modified silica gel that interacts predominantly with cis-diol sugars. ‘All of a sudden you can separate a cis- versus a trans-containing diol in a sugar by miles,’ she says.

Once separated, Wendlandt explains that analysing the products using typical analytical techniques – including NMR and x-ray diffraction – helps confirm whether the reaction was successful at inverting stereochemistry.
Sugars are not, however, the only target for Wendlandt. Her group has also developed similar techniques to invert the stereochemistry of tertiary carbon centres using a polyanionic tungstate photocatalyst, as well as ways to form higher energy alkene regioisomers.3,4
Probing mechanisms
‘The sort of standards of mechanistic analysis evolved around thermal catalysis,’ says Wendlandt, explaining that the team initially tried to understand these photo-catalysed reactions in the same way. ‘Very quickly, all of that fell apart for us.’
‘Most of that I think is because [these reactions] are often light-limited… so the only thing you see when you look into the reaction profile is light dependence. All this cool chemistry is happening and you’re kinetically blind to it.’
As such, understanding what’s happening in these short-lived excited states is challenging, making it hard to probe how substrates specifically invert to one stereocentre over another. ‘I think what has been important is being open to bringing in new technology,’ she adds, which has included collaborating with her colleagues to run some DFT calculations of the excited states in these reactions.
‘My lab has also recently started looking into the nanosecond timescale [by using] transient absorption spectrometry,’ Wendlandt says. Also known as flash photolysis, this technique measures changes in the absorption of a sample after it has been excited by an extremely short pulse of light. ‘We’re actually now able to get time-resolved data about the excited states and how those are interacting with the substrates,’ she explains.
In molecules where there are numerous chiral centres, like carbohydrates, these photocatalytic reactions seemingly target specific sites. However, Wendlandt explains that ‘these reactions [with sugars] aren’t site selective at all – they’re product selective’.5 She adds that when there are many possible reactions between isomers, ‘there are these hidden correlations between rate constants within the networks [of possible reactions] that can lead to the emergence of selective behaviour.’ This is unlike the conversion between only two isomers, where there is simply a kinetic and a thermodynamic product.
She hopes that understanding how changes in reaction conditions or catalyst influences rate constants will help the team design catalysts that more selectively form the desired isomer, be that the contra‑thermodynamic product or not . The team is also trying to make its catalysts more robust to improve the turnover number – the number of substrate molecules converted into product per catalyst over a given amount of time. Wendlandt adds that these contra-thermodynamic editing tools are ‘valuable because it’s not how chemistry works’. ‘All other chemistry flows downhill.’
References
1 Y Wang, H M Carder and A E Wendlandt, Nature, 2020, 578, 403 (DOI: 10.1038/s41586-020-1937-1)
2 K Suzuki et al, Sci. Rep., 2023, 13 (DOI: 10.1038/s41598-023-46796-4)
3 Y-A Zhang et al, Science, 2022, 378, 383 (DOI: 10.1126/science.add6852)
4 G Occhialini, V Palani and A E Wendlandt, J. Am. Chem. Soc., 2021, 144, 145 (DOI: 10.1021/jacs.1c12043)
5 H M Carder et al, Science, 2024, 385, 456 (DOI: 10.1126/science.adp2447)
Using light to push stereochemistry uphill

While chemistry usually follows the downhill pull of thermodynamics, Alison Wendlandt is creating higher‑energy stereoisomers in the final stages of synthesis
 Currently
reading
Currently
reading
Using light to push stereochemistry uphill
- 2
















No comments yet