How chemists are using light, enzymes and mechanical force to access higher‑energy stereoisomers without rebuilding a molecule from scratch
An organic molecule’s three-dimensional shape often dictates how chemists build it, forcing them to lock in stereochemistry at the same time as forming new bonds. But this can box chemists into using challenging, low-yielding or resource-intensive reactions.
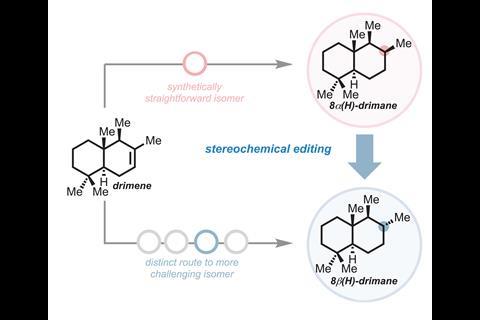
Stereochemical editing is an alternative approach. Here, chemists make late-stage alterations to the 3D arrangement of atoms, for example, by inverting specific stereocentres. This widens the synthetic routes to a given enantiomer, rather than following a linear pathway. Such stereochemical editing can unlock previously hard-to-access stereoisomers or simplify the route to a given enantiomer.
A guide to the terminology used in this piece:
Chiral: A molecule is said to be chiral if that molecule and its mirror image are non-superimposable.
Diastereoisomers: Stereoisomers are either enantiomers or diastereoisomers. Chiral molecules that have the relationship of mirror images are called enantiomers. All other stereoisomers are diastereoisomers including E/Z-isomers.
Enantiomer: Chiral molecules are mirror images of one another are called enantiomers. All other stereoisomers are diastereoisomers.
Epimer: Diastereoisomers that differ in only one configuration of two or more chiral elements.
Regioselective: A regioselective reaction is one in which one direction of bond making or breaking occurs preferentially over all other possible directions.
Stereocentre: Any atom in a molecule such that the interchange of two of its substituent groups leads to the formation of a different stereoisomer.
Stereoisomers: Compounds that exhibit the same constitution (the same molecular formula and connectivity of atoms) but different spatial arrangements. Stereoisomers can be further classified as enantiomers and diastereoisomers.
Stereochemistry: The study of the three-dimensional structure of molecules.
These tools can also offer a faster way to create to collections of molecules with subtle 3D differences, removing the need to synthesise each stereoisomer from scratch. Chemists can then test how these tweaks impact a molecule’s properties more quickly, which may be useful in drug development, since a molecule’s shape influences how well it fits into a receptor or enzyme.
Defying thermodynamics
Many stereochemical editing reactions will lead to a product that is more stable – or lower in energy. ‘If you’re breaking bonds and reforming bonds… and those are happening through microscopically identical elementary steps, then the system will necessarily descend into a thermodynamic control regime,’ explains Alison Wendlandt, an organic chemist developing stereochemical editing techniques at Massachusetts Institute of Technology in the US.
She adds that if, however, the reaction happens under kinetic control, this can lead to products that are higher in energy, or ‘contra-thermodynamic’.
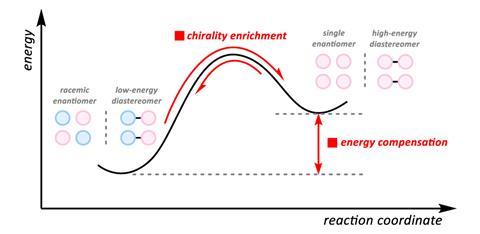
Converting a racemic mixture into a single enantiomer is inherently contra-thermodynamic as it leads to a decrease in entropy. Epimerisation – where one chiral centre in a molecule with at least two stereocentres changes configuration – can be contra-thermodynamic if the product is higher in energy than the starting material. Unfavourable steric clashes, or the loss of beneficial interactions with other groups or molecules, can result in a higher‑energy product.
How it’s done
There are several ways to do these types of stereochemical editing reactions, but whatever the strategy, a source of energy overcomes the barrier to reach the higher energy or ‘contra-thermodynamic’ product.
Wendlandt explains that her team tends to use photocatalysts. Light electronically excites the catalyst, which transfers energy to a substrate. This can weaken certain bonds that, for example, then allows the photocatalyst to abstract a hydrogen atom on a chiral carbon centre. As the substrate collapses back to the ground state, the photocatalyst returns the hydrogen back to the opposite face of the planar radical, generating the alternative epimer. Careful catalyst design ensures the reaction favours the higher-energy product over the original starting material, and can also improve regioselectivity. The team has so far used this approach to epimerise alcohol groups in carbohydrates and to invert the stereochemistry of tertiary carbon centres.
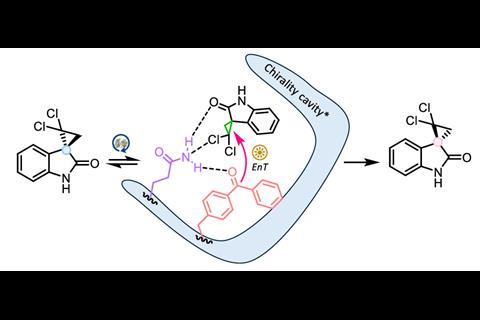
Enzymes offer another way to selectively synthesise higher-energy stereoisomers. These biological catalysts are inherently chiral, with active sites that can be engineered to favour one chiral substrate over another. Tailored side groups then drive the interconversion of chiral centres, for example, by opening and closing a chiral cyclopropane ring, pushing the reaction toward the higher-energy enantiomer.
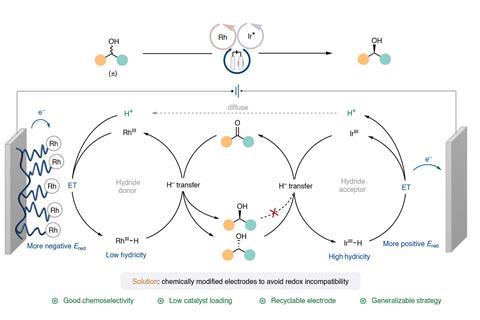
Transition metal catalysts can electrochemically convert a mixture of a pair of enantiomers into a single enantiomer. Using an iridium catalyst to first reduce a secondary alcohol into a ketone at the anode, with a rhodium catalyst then oxidising the ketone back into the alcohol at the cathode, is one approach. Fine-tuning the catalyst helps selectively reduce only one of the enantiomers, yielding an enantiomerically pure product.
Mechanical energy can also convert a mixture of enantiomers. For example, adding an amine base and glass beads to a racemate of chiral imines has been shown to drive the formation of one enantiomer over the other. Beads break down the crystals in the racemate into individual molecules, forcing them into solution. The enantiomers are then able to interconvert, which they were unable to do in the solid state. As the equilibrium between enantiomers favours one substrate over the other, this means that over time the amount of the more stable enantiomer increases, until it is the only enantiomer present in solution. This enantiomer can then re-crystalise once the reaction stops.
Contra-thermodynamic editing is about making precise changes to the spatial arrangement of existing chiral centres – converting stable configurations into higher-energy ones . The result is access to rare stereoisomers, complex sugar derivatives and other compounds that traditional synthesis struggles to reac
References
H Bai et al, J. Org. Chem., 2025, 90, 49, 17223 (DOI: 10.1021/acs.joc.5c02446)
Using light to push stereochemistry uphill

While chemistry usually follows the downhill pull of thermodynamics, Alison Wendlandt is creating higher‑energy stereoisomers in the final stages of synthesis
- 1
 Currently
reading
Currently
reading
Contra-thermodynamic stereochemical editing explained
















1 Reader's comment