By combining a silver catalyst with a chiral sulfur reagent, researchers are now able to stereoselectively aminate unactivated C–H bonds, allowing the team to make late-stage modifications to alkanes and drug-like molecules.
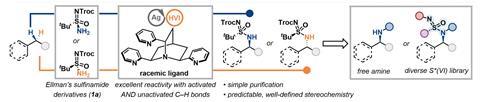
‘Previous methods for C–H amination mostly focused on substrates with activated C–H bonds,’ says Tuan Anh Trinh at the University of Wisconsin-Madison in the US, adding that these are often unable to aminate C–H bonds in alkanes, for example. Such techniques also typically require harsh reaction conditions, excess substrate or expensive metals like rhodium.
Trinh and the team have now developed a method that uses a silver catalyst and a chiral sulfinamide-type reagent to target unactivated C–H bonds. This produces a sulfonimidamide – a sulfonamide with an additional nitrogen-containing group. Reacting with triflic acid then removes the sulfonamide to generate a free amine. Using the method, the team were able to aminate a range of alkane and benzylic C–H bonds, as well as steroids or diterpenes. The team also found that the chirality of the sulfur(VI) reagent influenced which diastereomer formed.
‘Silver is not usually the first metal people think of [for C–H amination],’ says Derek Hu, part of the Wisconsin-Madison team. ‘The interesting thing about silver that differentiates it from a lot of the other metals is it’s really flexible in terms of what complexes it makes,’ he says. He adds that this provides ‘a lot of room for fine-tuning reactivity and selectivity by varying the ligands’.
Screening various nitrogen-containing ligands revealed that a quinuclidine compound with pyridine substituents led to the highest yields and produced a nearly equal ratio of both diastereomers. ‘I think that ligand hits a very sweet spot of both rigidity and bulkiness in order to control the selectivity of the reaction,’ says Trinh.
‘A key next question concerns the catalytic mechanism: what exactly is the silver–nitrogen intermediate,’ says Peter Zhang at Boston College in the US, who was not involved in the work. ‘Answering this question will be important not only for understanding this system, but also for designing the next generation of asymmetric C–H amination catalysts.’
The Wisconsin-Madison team was also able to show that the sulfonimidamide intermediate could be functionalised with various groups, including alkyne, pyridine and cyanide. Hu says that ‘sulfonimidamides are known to be medicinally useful’ so more ways of accessing that group ‘could be really useful in the [drug] industry’.
‘This is not just another example of C–H amination,’ says Zhang. ‘It is a platform approach that connects late-stage functionalisation, alkane feedstock upgrading and stereochemically defined amine synthesis.’
‘One broader lesson from this work is the power of ligand design,’ says Zhang. He adds that ‘with the right ligand engineering’ an underexplored metal like silver ‘can become the basis of a highly effective catalytic platform’.
References
T Anh Trinh et al, Science, 2026, 392, 421 (DOI: 10.1126/science.aee3321)















No comments yet