The synthesis of methanol from carbon dioxide can be dramatically improved by switching the catalyst support from zirconia to hafnia. Hafnia’s monoclinic crystal structure and insulating electronic properties efficiently stabilise single atoms of the active indium catalyst, boosting activity by up to 70% and substantially reducing the required loading of this expensive metal. These findings demonstrate the importance of carrier engineering and open the door to an alternative approach to catalyst design, say the authors.
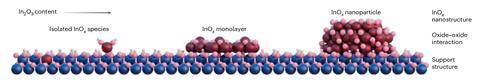
Green methanol, produced via the hydrogenation of carbon dioxide, is an important step in the decarbonisation of chemical and fuel production. Current commercial catalysts are based on copper and zinc, but deliver modest selectivity and often suffer irreversible deactivation by water. Over the last 10 years, indium oxides have emerged as a promising alternative, in particular when embedded on a monoclinic zirconia support. Various hypotheses have sought to explain the origin of this promotional effect, but the precise mechanism has remained elusive, hampering efforts to reproduce or improve upon this activity.
Other crystal structures of zirconium oxide produce a much smaller boost in catalytic activity and Javier Pérez-Ramírez and Sharon Mitchell at ETH Zurich therefore proposed that it was specifically the monoclinic form which boosted catalytic performance. However, a quick search of the materials database revealed only one accessible analogue: hafnia. Although typically considered catalytically inert, hafnia shares many properties with zirconia – notably, a wide band gap and high dielectric constant – which Pérez-Ramírez and Mitchell suspected would be crucial to the oxides’ auxiliary catalytic effect.
The team prepared a series of nanostructured indium–hafnium oxide catalysts, comparing their hydrogenation performance against the zirconia benchmark. The alternative support outperformed this standard across the board with the most significant improvement – a 70% boost in indium utilisation – achieved for single-atom catalyst loading.
These findings supported the initial hypothesis that the monoclinic structure and corresponding electronic properties were responsible for the observed activity enhancement and the team next performed a series of computational analyses to corroborate this theory further.
Their analysis suggests the monoclinic structure promotes the formation of oxygen vacancies around the indium centre, providing an accessible pocket in which the reaction can occur. The oxides’ insulating properties also help ensure the reaction remains localised around these active sites. ‘Materials with a wide band gap do not transfer electrons, so they are able to store protons and hydrides on the surface efficiently,’ explains Pérez-Ramírez. ‘The indium helps splitting the hydrogen, and the support stabilises very well these charged hydrogen species.’ Meanwhile, the hydroxylated surface of the hafnium oxide further stabilises the indium atoms in their most active form, increasing the lifetime of the catalyst.
‘Effectively, zirconia and hafnia have properties that go in a similar direction. But in the case of hafnia, the bandgap is slightly larger, the dielectric constant is slightly larger, and it is also more stable in the monoclinic lattice,’ says Mitchell. ‘All of these factors combined mean that effectively it increases the utilisation of the indium.’ This effect even worked with other methanol synthesis catalysts, with both zinc and gallium systems seeing big increases in activity, relative to the bulk material.
The team’s detailed analysis, combined with the wider implications of the carrier design particularly impressed Kelly Kousi, who leads a sustainable catalysis group at the University of Surrey. ‘The authors do not limit their findings to a single catalyst formulation. This really strengthens the importance of the research and the broader applicability of their design strategy,’ she says. ‘More importantly, the work seems to represent more than an incremental improvement to one catalyst, making it a meaningful step forward for heterogeneous catalysis as a whole.’
While the high cost of hafnium oxide means it is unlikely to ever become a commercial support, the real impact, and the focus of future work, will be in the intentional design of carrier structures with tailored electronic properties, says Pérez-Ramírez. ‘I think we have to elevate the carrier engineering to the same level as the active metal. It’s clearly a cooperative mechanism and carriers are much cheaper than the elements used in catalysis.’
Kelly Kousi’s affiliation was updated on 10 March 2026.
References
Y-T Chiang et al, Nat. Nanotechnol., 2026, DOI: 10.1038/s41565-026-02135-y















No comments yet