Can you make a catalyst that has the stability and recyclability of a heterogeneous one with the selectivity of a homogeneous one? Rachel Brazil talks to the scientists finding out
Catalysts make modern industrial chemistry possible. They are the mainstay of the petrochemical and pharmaceutical industries, allowing reactions to occur faster, cheaper and sometimes greener. And they’ve been around a long time: Swedish chemist Jöns Jacob Berzelius first used the term catalysis in 1836. He used it to describe a wide range of observed phenomena, both homogeneous systems – where the catalyst is in the same phase as the reactants – and heterogeneous systems, where the phase differs. Berzelius recognised that common chemical principles were involved, but the two branches of catalysis have developed independently and offer the chemical industry different strengths and weaknesses. Today, chemists are bringing these fields together to get the best of both worlds.
By the 1900s, all catalysts had been defined as substances that accelerate chemical reactions without appearing in the end products, but it later became clear that homogeneous and heterogeneous catalysts worked through different mechanisms and this has kept the fields largely distinct. ‘They don’t talk to each other because they developed as totally separate fields,’ says Gabor Somorjai, a veteran of catalysis science at the University of California, Berkeley, in the US. ‘Until recently they were taught separately, used separately and they are served by different literatures.’
One of the earliest examples of heterogeneous catalysis was the observation that red-hot metals such as iron or platinum could catalyse the decomposition of ammonia, recorded in 1813 by French chemist Louis Jacques Thenard. Heterogeneous catalysts, often metals, became widely used in industry, favoured for their easy recovery, so that the expensive catalyst can be removed from the product and re-used.
They don’t talk to each other because they developed as totally separate fields
Gabor Somorjai, UC Berkeley
The first industrial homogeneous catalyst was used as part of the lead chamber process for making sulfuric acid. In 1746, Birmingham-based British industrialist John Roebuck exploited the catalytic effect of nitrogen oxide gases on the reaction between moist sulfur dioxide and air. Homogeneous catalysts based on transition metals are now more common, such as the organorhodium compound used industrially in hydroformylation – the production of aldehydes from alkenes. The metal ions in such systems can catalyse reactions due to their variable oxidation states. Being surrounded by organic ligands also provides a very specific steric environment which will favour a specific product. Changing these ligands allows chemists to tune the catalyst’s selectivity.
‘Generally homogeneous catalysts are very selective but not very active and not very stable, whereas heterogeneous catalysts are pretty active (you can run them at higher temperatures as they are more stable) but not very selective.’ explains Charles Sykes, a surface chemist at Tufts University in Massachusetts, US. One gives you selectivity; the other, activity and ease of separation. But is there a way to combine the approaches? Can hybrid catalysts bridge the heterogeneous–homogeneous gap?
Catalyst in the trees
Attempts to find hybrid systems have had mixed success to date. This often involves ‘heterogenising’ homogeneous catalysts by covalently bonding them onto a porous support material such as silica. They can be directly chemisorbed or bonded to silanol groups via a linker molecule. Former industrial chemist Robert Farrauto is currently at Columbia University in New York, US, and has experience of this approach from his 37 years with the Engelhard Corporation and BASF. ‘We had marginal success, but invariably when you would bond the material, you would lose a bit of your performance because you had lost a degree of freedom with some of the [catalyst] binding sites now bonded to the substrate, rather than to the reactants,’ he explains. ‘In other cases you would also start seeing leaching occurring, and the [catalyst] material would begin to dissolve off the support.’
Today, researchers are trying to do better with various strategies to combine the properties of homogenous and heterogeneous catalysts. The goal is a highly reactive, stable catalyst with 100% product selectivity – and all at low cost, of course. And while the objective is shared, the heterogeneous and homogeneous catalyst communities are looking at the problem from their different perspectives.
When you bond a homogeneous catalyst to a support, you lose a bit of your performance because you lose a degree of freedom
Robert Farrauto, Columbia University
From the heterogeneous perspective, the objective has been to create smaller and more finely dispersed catalyst particles. Catalysts such as metal nanoparticles can be distributed within a porous support material, and improving their performance is linked to decreasing the nanoparticle size, to give more active catalytic sites. But this competes against the nanoparticles’ tendency to aggregate into clusters, which as well as being less efficient can also increase side reactions. The ultimate aim is to create single-atom catalysts – isolated catalytically active atomic sites dispersed within a support, which might mimic a homogeneous catalyst, providing similar reactivity and product selectivity.
Somorjai and collaborators at Berkeley came up with a new strategy to heterogenise homogeneous catalysts for the formation of cyclopropane from alkenes, which in industry requires a homogeneous catalyst. They have developed dendrimer-encapsulated metal nanoparticles immobilised onto mesoporous silica through hydrogen bonding. Dendrimers are tree-shaped branched polymers – in this case polyamidoamine – and they encapsulate gold, platinum, palladium or rhodium nanoparticles. ‘It turned out if I used [dendrimers] to put the 1nm [metal] clusters in, they got stuck in the branches and they stayed put,’ explains Somorjai. The dendrimer protects the clusters from aggregation or leaching. Plus the size of the clusters, generally around 40 atoms, are also small enough to electronically behave like discrete molecules and bind reactants more strongly.
Dendrimers also tune the catalytic properties of the system in a similar manner to the ligands surrounding homogeneous catalyst ions, and seem to provide the kind of chemical selectivity difficult to achieve with most heterogeneous catalysts. This includes stereoselectivity; for example, in the reaction between propargyl pivalate (C8H12O2) and styrene (C6H5CH=CH2), Somorjai used a dendrimer-encapsulated 2nm gold cluster catalyst to achieve a fivefold increase in cis-cyclopropane (over trans) compared to the same reaction with a gold chloride homogeneous catalyst. Smaller dendrimers produced greater steric preferences, due to the increase in the spatial restriction around the gold centres.1
Alloy allies
Another ‘halfway house’ approach is to create very dilute alloys that include catalytically active metals. This is the approach being developed by Sykes, in collaboration with colleagues at Tufts and at University College London in the UK. ‘We carefully pick the combinations: for example, platinum and copper, as platinum is very soluble in copper and would rather be surrounded by copper atoms than by platinum atoms, so the thermodynamics give you a “single-atom alloy”,’ he explains. Using scanning probe microscopy, they could visualise individual platinum atoms in the copper bulk.
The idea of using synergistic combinations of an expensive catalytic metal with a cheaper host to, for example, catalyse C–H bond activation or hydrogen dissociation reactions could be a money-saver (platinum costs around $30 per gram (£20) to copper’s one cent). And with platinum there is an added advantage that the alloys are not susceptible to ‘coking’, the deactivation of the catalyst due to side reactions that cause carbon deposition. ‘To break a carbon–carbon bond you need more than one platinum atom, and the fact that there are just single atoms means that you can’t break the bonds – so you don’t get a build-up of coke,’ explains Sykes.2

The alloys can also provide greater chemical selectivity. ‘We have been able to design heterogeneous catalysts with the atomic scale detail that homogeneous catalysts have had for 100 years, so we are now at the point where we can get the best of both worlds,’ says Sykes. Catalysis on metals generally occurs at defect sites in the surface structure, but here the combination of single atom catalytic sites surrounded by weakly binding copper sites provides an environment that can better control the types of reactions occurring.3
A similar approach from the lab of Peter Wasserscheid at the University of Erlangen–Nuremberg in Germany uses what they call ‘supported catalytically active liquid metal solutions’. These are droplets of a palladium–gallium alloy, with 50 times more gallium than palladium , supported on macroporous glass. The system was used to catalyse butane dehydrogenation. At the reaction temperature of 450ºC the alloy is a liquid, but the palladium exists on the liquid gallium surface as single isolated atoms that act as catalysts.4
Though heterogeneous catalysis is favoured in industry, it’s not always possible. For example, hydroformylation, the production of aldehydes from alkenes and syngas (hydrogen, carbon monoxide and carbon dioxide), is carried out on a million-ton scale worldwide. The reaction – an addition of a formyl group (CHO) and a hydrogen atom to a carbon–carbon double bond – is carried out with a homogeneous rhodium catalyst, as too many side reactions occur with heterogeneous catalysis. ‘Since it was invented in 1938, researchers in industry as well as academia have tried to heterogenise the reaction, but they weren’t successful,’ says Robert Franke from Evonik Performance Materials, based in Marl, Germany. His company is now trying to change that, with their supported ionic liquid phase (SILP) approach.

They have developed a system that uses a rhodium ion catalyst in an ionic liquid, a salt that exists in the liquid phase below 100°C, and in this case an imidazolium cation with a binary amine anion. The ionic liquid is physisorbed as a nanometre-thick layer on a porous support material, retaining catalytic performance when in contact with the gaseous reactants and products. ‘SILP technology is not simply heterogeneous, it is a unification of heterogeneous and homogeneous catalysis – because inside your support you still have the homogeneous catalyst and I think that is the real great advantage,’ explains Franke. ‘It has exactly the same selectivity that a homogeneous catalyst has.’ For example, using a diphosphite ligand attached to the rhodium catalyst, the SILP approach yielded 99% of the desired n-pentanal product.5
Evonik are testing the system in several mini plants as part of the EU-funded project Romeo. The aim is to reduce the energy consumption of hydroformylation by up to 80%, emissions by up to 90% and build a full-scale plant in the next three years. They still need to increase the lifetime of the catalyst, as currently it becomes deactivated after 3000 hours. Franke says they are looking for a lifetime of at least 8000 hours (around a year).
The electrocatalytic buzz
Away from the petrochemicals industry, catalysts also play a huge role in electochemical reactions. Electrocatalysts are crucial for speeding up the reactions in fuel cells, batteries and solar cells. Yogesh Surendranath, a chemist at the Massachusetts Institute of Technology in the US, has developed a method of electrocatalysis that draws from both heterogeneous and homogeneous catalysis. He has bonded catalyst molecules to graphite electrodes using a pyrazine ring, creating a conjugated aromatic linkage between the catalyst metal centre and the electrode surface. The result is a radically different mode of catalysis with significantly higher activity than the equivalent catalysts in solution – a stark contrast to the usual situation, where supported catalysts have inferior performance.
Surendranath has made these graphite-conjugated catalysts with rhenium coordination compounds for the selective reduction of CO2 to CO.6 ‘In principle, any homogeneous catalyst – provided it can be modified in the appropriate way – should be amenable to the chemistry that we’ve developed,’ he says. The conjugation dramatically alters the catalyst’s electrochemical behaviour compared to solution phase or other surface-tethered approaches. ‘Typically, when when people modify an electrode surface with a molecule, they are simply anchoring the molecule to the surface. What we are doing is electronically connecting the molecule in a way that permits strong orbital interactions between the molecule and the electronic band states of the metallic solid,’ explains Surendranath,
Evidence for a different type of catalysis also comes from measurements of the oxidation state of the transition metal ion in the catalyst – it remains unchanged. The catalyst is not behaving like a conventional homogeneous molecular catalyst but more like the metallic active sites exploited in heterogeneous catalysts. ‘We still get single-site activity for the surface sites, but the way in which they do catalysis is very different because of their altered electronic structure,’ says Surendranath. ‘Ours is a hybrid of electronic structures just as much as it is a hybrid of homogeneous and heterogeneous catalysis.’
Combining the properties of tuneable molecular catalysts with the electronic properties of metallic extended solids could be useful outside electrocatalysis alone, Surendranath suggests. ‘We can exploit new mechanisms to do reactions that may otherwise not be possible with a traditional heterogeneous catalyst, or with a molecular catalyst dissolved in solution.’
So which of these approaches is likely to provide the next generation of industrial catalysts? ‘I’m absolutely sure that the future of catalysis will involve catalytic systems which bridge the gap between both technologies,’ says Franke. ‘If it’s SILP or some other kind of technology to heterogenise [homogeneous catalysts], I couldn’t judge.’ Of his own technology, Surendranath says ‘It’s hard to say what the impact will be on an industrial scale. The idea that any given catalyst platform is a panacea in catalysis is definitely not appropriate.’
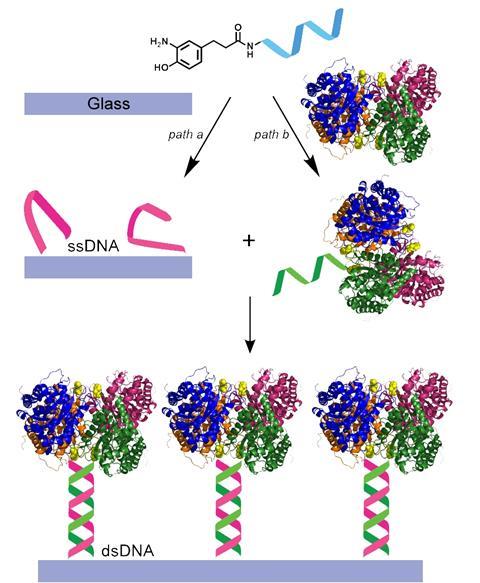
Somoraji has now turned his attention in another direction – to enzymes, nature’s own catalysts. Isolated enzymes have been used in detergents since 1914 and are widely used in the food and fine chemicals industries. Somoraji is now trying to heterogenise them, while retaining their function. With Berkeley collaborator Matthew Francis, he has used single strands of DNA to immobilise enzymes to a glass surface. Complimentary DNA strands are attached to the glass surface and the enzyme’s N-terminus, allowing the enzyme molecules to be arranged in a particular orientation to retain their catalytic activity.7
For a long time catalysis has relied on serendipity or combinatorial approaches to find new materials, but chemists are now moving towards more rational design principles. ‘It’s really an exciting time right now, because we are understanding active sites with atomic scale resolution and then being able to model them with theory,’ says Skyes. The breakdown of the old heterogeneous and homogeneous divisions may be the result and lead to the development of faster, cheaper and greener hybrid catalysts.
Rachel Brazil is a science writer based in London, UK
References
1 E Gross et al, Nat. Chem., 2012, 4, 947 (DOI: 10.1038/nchem.1465)
2 M D Marcinkowski et al, Nat. Chem., 2018, 10, 325 (DOI: 10.1038/nchem.2915)
3 G Kyriakou et al. Science, 2012, 335, 1209 (DOI: 10.1126/science.1215864)
4 N Taccardi et al, Nat. Chem., 2017, 9, 862 (DOI: 10.1038/nchem.2822)
5 M Jakuttis et al, Angew. Chem. Int. Ed., 2011, 50, 4492 (DOI: 10.1002/anie.201007164)
6 M N Jackson et al, J. Am. Chem. Soc., 2018, 140, 1004 (DOI: 10.1021/jacs.7b10723)
7 K S Palla et al, J. Am. Chem. Soc., 2017, 139, 1967 (DOI: 10.1021/jacs.6b11716)












No comments yet