Andy Extance tells the overlooked story of crystallographer June Sutor, whose C–H⋯O bonding hypothesis was unjustly suppressed
It was probably following long weeks in the early 1960s analysing x-ray diffraction data that an idea ahead of its time crystallised in Dorothy June Sutor’s mind. Decoding the purine crystal structures the spots represented likely helped her imagine a previously inconceivable chemical phenomenon.
Many crystallographers at the time laboured through this intensive process, but also benefited from it, explains Maureen Julian, who became a close friend of Sutor’s a few years afterwards. ‘It promoted a much more in-depth study of each of the compounds, because it took so long to do,’ says Julian, who teaches crystallography at Virginia Tech in the US.

In 19621 and 19632, Sutor published two papers describing attractive hydrogen bonding interactions involving hydrogen atoms attached to carbon atoms, as sole author. Yet her ideas were rejected by Jerry Donohue, an American crystallographer and hydrogen bonding expert. And Donohue was powerful, in part from thanks to sharing an office with James Watson and Francis Crick in 1952, and his subsequent role in helping guess DNA’s double-helix structure.
‘This type of hydrogen bonding became almost unmentionable,’ wrote Carl Schwalbe, a retired crystallographer at Aston University in the UK, in his account of her story.3 Sutor’s evidence wasn’t conclusive, and her ideas would only be widely accepted as true after she left structural crystallography research. ‘Someone like Donohue, who had solved dozens of crystal structures, might conceivably resent June Sutor, who solved a handful and then dived into the literature with this new concept,’ Schwalbe tells Chemistry World. ‘Acceptance of women in science, particularly the physical sciences, was by no means complete,’ Schwalbe wrote.
Sutor was ‘very, very proud’ of her hydrogen bonding work according to Julian. She describes her friend as being brilliant, generous, caring and dignified, thanks partly to her upbringing. She was born in 1929 in New Zealand, the daughter of a ‘coach builder’, census records show. Sutor’s father imported heavy machinery from England and then assembled fire engines, buses, ambulances and other vehicles, according to Julian. The enterprise ‘was extremely successful’ and eventually ‘June’s father received a generous offer to buy the whole business’, Julian says, making the family wealthy.
Punching cards
Sutor gained an MSc and PhD from the University of Auckland in 1952 and 1954, supervised by Frederick Llewellyn, the first crystallographer in New Zealand. Her clear talent earned her first-class honours, and senior scholar and postgraduate scholar awards, propelling her into the ranks of crystallographers transforming chemistry. The growing field started in 1912, when German physicist Max von Laue and colleagues found atoms encoded patterns into X-rays passing through them, recorded as spots on photographic plates. The following year UK-based crystallography trailblazers Lawrence and William Bragg worked out how to mathematically decipher the encoded chemical structures.
By the 1940s many crystallographers were studying biomolecules, with Dorothy Hodgkin leading the way with cholesterol at the University of Oxford in the UK. Sutor therefore came to England, where she got a second PhD from Cambridge, publishing structures of purine compounds, including caffeine in 1958. After working in Australia for a year, Sutor moved to lecture at Birkbeck College in London, where John Bernal, Rosalind Franklin and Aaron Klug pioneered x-ray crystallography in molecular biology. This was where she wrote the 1962 and 1963 papers. Sutor was ‘a modest and highly competent scientist’ says Olga Kennard, who knew Sutor slightly and founded the UK-based Cambridge Crystallographic Data Centre (CCDC), but is now retired.
Woe betide you if you drop the stack of punch cards, because there’s no way of putting them back in the right order
Scientists in those days used cameras that let them reproducibly locate spots made on films by x-ray diffraction patterns. Schwalbe’s first crystal structure was a boron hydride. His Weissenberg camera gave festoons, or chains, containing around 9000 spots, which he indexed and assigned intensities for by hand. ‘You’d hold this calibrated strip next to each one, and note which point on the strip gave the closest match in terms of the degree of blackness,’ Schwalbe recalls. Often male crystallographers ‘would put their wives to work doing a lot of this’, he adds.
Calculating structures from such indices initially took months by hand, until scientists got mechanical punch-card tabulators like the one Hodgkin used to get a structure for penicillin in 1945. By the time Sutor reached Birkbeck she could do her calculations ‘on Mercury in the University of London’, she wrote in correspondence held in the UK’s National Archives. She was perhaps referring to an early Ferranti Mercury computer. In a later CV, also in the National Archives, she declares an interest ‘in electronic computing and has written programs for the EDSACs in Cambridge and Mercury in London’.
Judith Howard, a crystallographer at the University of Durham in the UK, recalls still translating code produced by one machine that put holes in tape into punch-cards in the 1970s. ‘If you had a really large dataset translated into a stack of cards, woe betide you if you drop them, because there’s no way of putting them back in the right order.’
Acceptable atoms?
Crystallography both relies on and helps improve scientific understanding of interactions between atoms whose roots lay in the late 19th century work of Johannes van der Waals. His many findings included limits to how close two atoms can approach each other: the van der Waals radius. This crucial parameter for crystallography is considered as half the distance between the closest approach of two non-bonded atoms of a given element.
Sutor exploited that concept together with the idea of electronegativity that Linus Pauling described in 1932. Electronegative elements accumulate electrons in higher density around their nuclei than other atoms they’re covalently bonded to, giving them a partial negative charge. The less electronegative atoms that they’re bonded to therefore have fewer electrons, and partial positive charges.
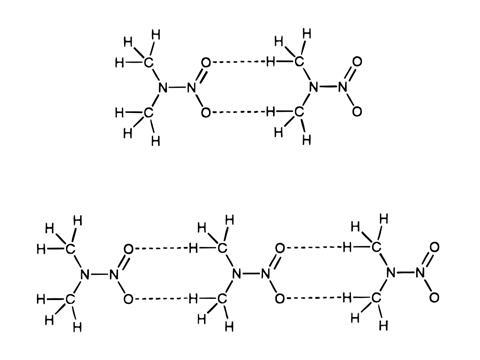
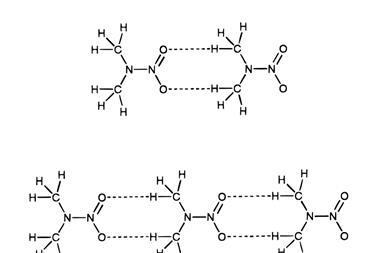
In hydrogen bonds, these partial charges interact to help orient an electronegative ‘donor’ atom, covalently bonded to a hydrogen atom, which non-covalently attracts another electronegative ‘acceptor’ atom. The classic example is between O–H bonds and oxygen atoms in water. Carbon and hydrogen have very similar electronegativity values, making it seem unlikely that carbon could be a donor.
‘Hydrogen bonding involves a shortening of the van der Waals distance between the atoms concerned,’ Sutor wrote in her 1963 paper. ‘The hydrogen atom lies between two electronegative atoms, and it is assumed that the donor, hydrogen, and acceptor atoms are aligned linearly. The donor and the acceptor atom are usually the most electronegative of the elements – fluorine, oxygen, nitrogen, and chlorine – but a C–H group that is activated by groups promoting ionisation or partial ionisation can also take part in hydrogen-bond formation.’
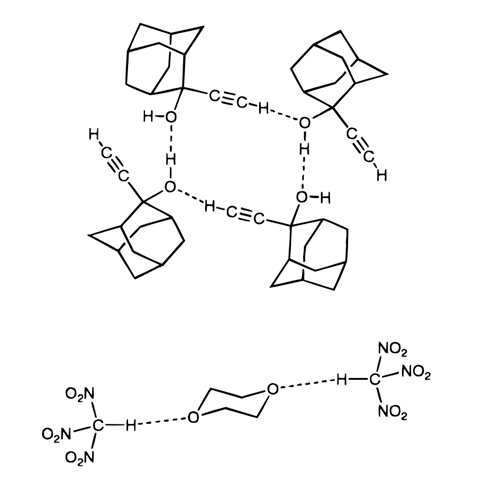
That final assertion was bold, but there was supporting evidence. For example, halogenated hydrocarbons like chloroform dissolve better than expected in solvents that contain oxygen or nitrogen atoms. Some scientists attributed that to hydrogen bonding involving a hydrogen atom attached directly to chloroform’s single carbon atom. Similarly, spectroscopy hinted at C–H⋯O hydrogen bonds – but only crystallography could show precise atomic arrangements.
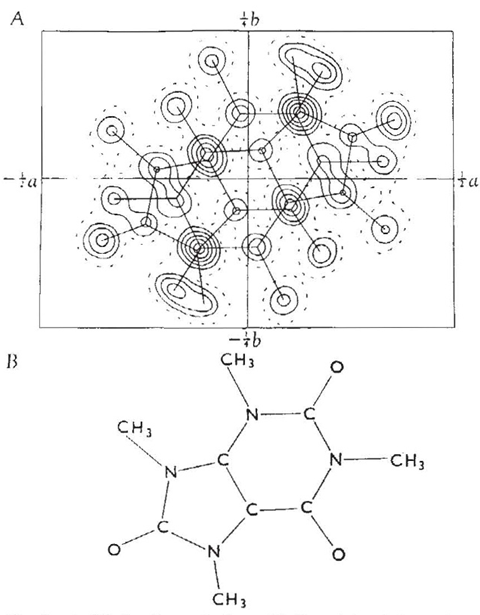
One clue is the distance between a methyl group and an oxygen in Sutor’s crystal structure of 1,3,7,9-tetramethyluric acid, or theacrine.4 Using contemporary van der Waals radii, the distance between the methyl carbon and oxygen atoms should have been 3.40 ångstroms (Å; 1Å = 0.1nm). Sutor measured the distance at just 3.00Å and estimated that this equated to 2.26Å between the methyl hydrogen and the oxygen. She based her 1962 paper on eight such purine crystal structures with carbon–oxygen distances below 3.30Å.
Sutor’s follow-up paper included 24 similar published examples, including DNA, and involved interactions with hydrogen atoms bonded to carbon in various environments. ‘It is tempting to speculate as to whether the C–H⋯O hydrogen bond plays as important a part in the structure of biological molecules as the other kinds of hydrogen bonds do,’ she wrote.
Philip Coppens at Brookhaven National Laboratory in New York, US, provided further evidence in 1964: a neutron diffraction study of 2-nitrobenzaldehyde. The molecule is well set up to form an intramolecular C–H⋯O hydrogen bond because the aldehyde hydrogen is adjacent to the nitro group’s oxygen atoms. Also, hydrogen atoms show up more clearly with neutrons than with x-rays, because neutrons’ interaction with atoms is not dependent on atomic number, unlike x-rays. Coppens described a short, 2.38Å, oxygen–hydrogen contact. But there was a problem: the molecule’s aldehyde and nitro groups didn’t lie in exactly the same plane as the aromatic ring.
‘You would expect for maximum electron overlap that the aldehyde and the nitro groups would be as co-planar as they could be,’ Schwalbe says. ‘What is stopping them? Steric hindrance. What might overcome the steric hindrance? Hydrogen bonding. Steric hindrance prevents them becoming completely coplanar, but they are perhaps a bit closer to coplanarity than they would be.’
Hydrogen bond villain?
The main critic was Jerry Donohue from the University of Southern California, who had published an influential review on hydrogen bonding in 1952. He had also told Watson and Crick that they were using the wrong structures for DNA’s thymine and guanine bases, affecting their hydrogen bonding. In 1968, Donohue wrote a book chapter entitled The C–H⋯O Hydrogen Bond: What Is It? It ruled out several of Sutor’s samples due to data problems, but added seven further examples of short C–H⋯O interactions. These included Coppens’s study, with Donohue highlighting 2-nitrobenzaldehyde’s refusal to lie in a flat plane. He argued that this result showed that ‘an O–H distance of 2.38Å is not presumptive evidence that hydrogen-bond formation is occurring’.
Donohue also argued that Sutor overestimated the van der Waals distance between hydrogen and oxygen atoms. That overestimate was more likely to be the reason Sutor was seeing shorter distances in her measurements, he implied. What she said were hydrogen bonds therefore involved no attractive interactions at all, Donohue reasoned. Considering the data then available, he asserted that oxygen–hydrogen contacts down to 2.20Å could occur without hydrogen bonding. ‘The answer to the question in the title of this section is “It isn’t,”’ Donohue concluded.
Because it was a book chapter this criticism was more difficult to rebut than if it had been a standard research paper, Schwalbe points out. Robin Taylor, who is also a retired CCDC chemist, adds that the book was on the shelves of every crystallography lab at the time. ‘Basically he was scathing about Sutor’s suggestion,’ Taylor says. ‘It is rumoured that anyone who wrote a crystal structure paper and posited the existence of C–H⋯O bonds got it refereed by Donohue and it was rejected.’
After her last C–H⋯O paper was published, the funding for Sutor’s position ran out and she moved back to her native New Zealand. According to Julian, after working at the country’s Department of Scientific and Industrial Research, Sutor left to look after her father, who was suffering from cancer and died in 1966. While wealthy enough to ‘set her own priories’, Julian says Sutor then became desperate to return to research.
Sutor was lucky to find a job opening at University College London, working with Kathleen Lonsdale, the eminent crystallographer who started her career working with the Braggs. Lonsdale hired Sutor in 1966 to apply crystallography to study how urinary calculi – bladder stones – formed and how to prevent them. Lonsdale’s papers – including correspondence with Sutor – are held in the UK’s National Archives and were an invaluable resource for this article.
Lonsdale needed a researcher already expert in crystallography, but Sutor had other skills too. ‘Dr Sutor has shown herself to be particularly good at obtaining the necessary contacts with and cooperation from senior hospital staff,’ Lonsdale wrote to J W McAnuff, assistant director of The Nuffield Foundation. So, around the time Donohue published his criticism, Sutor was analysing French emperor Napoleon III’s bladder stone.5
In 1969 Lonsdale secured Sutor a two-year £10,000 Nuffield grant, with her proposed annual salary for the period being £2,600. Lonsdale and Sutor also wrote a book on calculi together, but publishers’ responses were predictably stony. Sutor persisted after Lonsdale’s death in 1971, trying and failing to get it published until 1974. Sutor continued to study the stones, noting in a 1981 paper that since she had become partially sighted in 1979, she had ‘become interested in theoretical aspects of stone growth’.
It’s easy to imagine that Sutor’s shift away from structural crystallography might have been a response to the rejection of her C–H⋯O bond hypothesis. It’s true that the only time Sutor mentioned C–H⋯O bonds in the correspondence between her and Lonsdale was in her CV. But Julian, who worked in the Lonsdale group from 1966–1968, remembers Sutor giving a seminar on the subject and emphasising the high number of citations her papers had attracted.
Julian highlights the connection between Donohue, Watson and Crick, and the way the latter pair treated Rosalind Franklin. In 1983 Julian published a short biography of Franklin, describing how Watson and Crick had used ‘shady behaviour’ to access crystallographic data from DNA that Franklin had collected.6 In that account, Julian mentions the ‘men’s club’ atmosphere of King’s College London, where Franklin worked before moving to Birkbeck. Now, she suggests Donohue’s attitude to Sutor may have similar origins. However, Kennard argues that ‘the opposition to her theory was in no way related to June being a woman’. ‘The crystallographic community was always very supportive of women scientists,’ she says.
Nevertheless Sutor’s work became taboo. Consequently, Indian crystallographer Gautam Desiraju didn’t consider such interactions during his PhD studies at the University of Illinois at Urbana-Champaign, US, in 1975 ‘even though I was practically looking at them’. Desiraju, now at the Indian Institute of Science in Bangalore, first explored C–H⋯O bonds in the mid-1980s, when encountering crystal structures that couldn’t be explained without them. In the years since, he’s learned just how fierce Donohue’s opposition was. ‘I talked to my colleagues in the US, and they said that they were scared to use the words “C–H⋯O hydrogen bonds” because Donohue was so influential,’ Desiraju says. ‘They said, “Look, it’s a question of getting my grant.”’
Poignant legacy
Desiraju could start work on the topic largely thanks to a 1982 paper by Taylor and Kennard that rescued Sutor’s C–H⋯O bonding claims.7 Analysing 113 neutron diffraction patterns in the Cambridge Structural Database, they found a statistically significant tendency favouring short intermolecular C–H⋯O contacts over C–H⋯C or C–H⋯H. ‘We had a hell of a lot more data,’ Taylor recalls. ‘That was really the limitation of Sutor’s work. You can’t be sure with that quantity of data. Particularly, [in] most of those structures you couldn’t actually see the hydrogen atoms.’

Neither scientist that peer-reviewed the study rejected it, although Taylor remembers one ‘was distinctly grumpy’. ‘He said “I wouldn’t call them hydrogen bonds, I’d call them Sutors”. I would lay money that it was Donohue. It wasn’t, based upon the context, in any honorific sense. It was basically “This woman suggested them but frankly they’re just an invention”.’
Overall Sutor’s carbon–oxygen distances proved reliable, differing from modern measurements by 0.03Å on average, with the worst being 0.07Å. Due to the difficulty of locating hydrogen atoms with x-rays, the average discrepancy in hydrogen–oxygen distances from modern values is 0.12Å and up to 0.26Å. On the whole the cases that Sutor cited still support her conclusion, except when the hydrogen atoms hadn’t been located in the structural analysis.
Although Sutor survived to see her vindication by Taylor and Kennard, there’s no indication of how she felt about it and she never mentioned it to Julian. Sutor died of cancer, like her father, on 27 May 1990. She left an estate of £536,210 (worth over £1 million today), according to probate records, but electoral roll records show no immediate family surviving her. Instead, her will established June Sutor Fellowships for blindness prevention research at Moorfields Eye Hospital in London in 1993, which are still active today. But Sutor’s hypothesis about C–H⋯O hydrogen bonding is an important chemical legacy in inspiring scientists to look for other similar weakly attractive interactions. ‘There are now dozens of them,’ says Taylor.
Desiraju would go on to write a book on weak hydrogen bonding interactions, and dedicate a 1996 review on C–H⋯O hydrogen bonding to Sutor.8 That was out of respect to her systematic approach, but he also ‘felt that everybody had sort of forgotten her by then’. ‘Now it’s impossible to think about structural chemistry or structural biology without C–H⋯O,’ he adds.
Meanwhile, Desiraju calls Donohue’s apparent actions ‘a very sad aspect’ of Sutor’s story. ‘When a big scientist goes wrong, it can have a very debilitating effect on a field,’ Desiraju says. ‘I think prominent scientists should therefore be very careful when they criticise a piece of work.’
Andy Extance is a science writer based in Exeter, UK.
Sutor’s funeral was held at All Saints Church, Highgate in July 1990. The order of service speaks of both her scientific career and her character.
’What was also apparent was her great strength of character: her determination to succeed in what is often regarded – or used to be – as a man’s world – the world of physics and chemistry, and scientific research.’
Click the image to download the complete order of service.
References
1 D J Sutor, Nature, 1962, 195, 68 (DOI: 10.1038/195068a0)
2 D J Sutor, J. Chem. Soc., 1963, 1105 (DOI: 10.1039/JR9630001105)
3 C H Schwalbe, Crystallogr. Rev., 2012, 18, 191 (DOI: 10.1080/0889311X.2012.674945)
4 D J Sutor, Nature, 1960, 188, 47 (DOI: 10.1038/188047a0)
5 D J Sutor, Nature, 1968, 218, 295 (DOI: 10.1038/218295a0)
6 M M Julian, J. Chem. Educ., 1983, 60, 660 (DOI: 10.1021/ed060p660)
7 R Taylor and O Kennard, J. Am. Chem. Soc., 1982, 104, 5063 (DOI: 10.1021/ja00383a012)
8 G R Desiraju, Acc. Chem. Res., 1996, 29, 441 (DOI: 10.1021/ar950135n)












No comments yet