Super-resolution fluorescence microscopy earned three of its creators a Nobel prize this year. Emma Stoye focuses in on their story
Seeing is believing, as the old saying goes. It is one thing to appreciate that every living thing is made up of cells and molecules, but it is another to see it right in front of you. The brightly coloured blotches and wiggling outlines captured by microscopes have inspired many budding scientists over the years. And it seems they inspired this year’s chemistry Nobel committee too, as the prize was awarded to Stefan Hell, Eric Betzig and W E Moerner. These three pioneers of super-resolution microscopy made it possible to peer into living, breathing cells in unprecedented detail. Now, the methods based on their discoveries are being used all over the world to zoom in on the nano-universe and reveal its inner workings.
And it’s not just about making pretty pictures. ‘Ultimately every disease leads back to a disturbance of some kind on the cellular level,’ says Hell, who is based at the Max Planck Institute for Biophysical Chemistry in Göttingen, Germany. ‘And only once we are able to look into the cell will we be able to determine exactly what went wrong.’
For over a century, light microscopy – the go-to method for looking at living cells and tissues – could only be used to look at structures down to half the wavelength of light, or about 200nm, a limit recognised in the late 19th century by Ernst Abbe. While electron microscopy allows much higher resolution, it requires samples to be prepared at low temperature in a vacuum, and is therefore unsuitable for living things.
By challenging – and ultimately overcoming – Abbe’s limit, Hell, Betzig and Moerner opened the door to a whole new world of possibilities.
Backing the basics
For Hell, the initial suspicion that Abbe’s diffraction limit could be broken started while he was still a PhD student at the University of Heidelberg in Germany. The hunch continued to niggle in the back of his mind until, in the 1990s, he formed a more concrete idea of how it might be done. ‘I knew I had to get inside the molecules,’ says Hell. ‘I couldn’t do it by means of light – it would be impossible to change the light.’

His idea was that if you could find some way of turning off all but a tiny nanosized area of the light emitted by a source – such as a group of fluorescent molecules – you could get rid of light that would create blurriness, and achieve much higher resolution. After working as a postdoc in Finland, Hell returned to Germany to pursue this idea, and published his first theoretical proposal for how the limit could be overcome in the mid-1990s.
His brainchild was stimulated emission depletion (Sted) microscopy, which uses two laser sources – one narrow beam to excite the fluorescent molecules in a tiny spot, and a second beam shaped like a hollow tube to quench all the fluorescence surrounding that spot. The sample is then scanned one nanometre-wide strip at a time to build up a high resolution picture. It took another five years until Hell actually demonstrated Sted, using it to visualise spots of fluorescence far smaller than the conventional diffraction?limit.1
As with any research that challenges textbook ideas, his work attracted criticism from the wider scientific community. ‘First they said it wouldn’t work in principle,’ Hell remembers. ‘When it did work in principle, they said perhaps it will only work in a special case. When it worked in a special case, they said it won’t work in living organisms … and so on.’ But he didn’t allow himself to be fazed, and went on to prove the critics wrong every step of the way. ‘Because I knew that this idea was fundamentally correct, I held on to it and did not allow myself to be misled when people told me it wouldn’t work,’ he says. ‘There were certain points that had to be overcome and solved, but they were not basic, which is why I believed in it,’ he adds. ‘There is a big difference between having a technical problem and having a basic problem – the basic ones will probably remain; technical problems can be overcome.’

And that determination has certainly paid off. In the relatively short time since the first demonstration of Sted, the technique has been adopted by researchers from all over biomedicine. Sted microscopes – of which there are currently just a handful available worldwide – have recently been used to monitor the behaviour of actin fibres inside nerve cells, to track the movement of ions within contracting heart cells, and to study the organisation of DNA-binding proteins. And this year, Hell’s group published their latest design for a Sted microscope for biomedical research,2 capable of achieving resolution of 20nm.
‘It does not make sense any more … to have blurry images,’ says Hell. His group continue to improve Sted technology, working on pushing the boundaries of resolution still further, as well as collaborating with others to apply the techniques to different problems. ‘It is huge fun!’ he says.
Single shiners
While Hell was putting his theory together, Betzig and Moerner laid the foundation for a separate group of techniques that overcome Abbe’s limit. While Sted works by quenching the light emitted by glowing fluorescent molecules, these rely on controlling the behaviour of the molecules themselves. One of the first breakthroughs was when Moerner became the first person to detect a single fluorescent molecule of pentacene in 1991, using laser-based techniques to measure the light it absorbed.3
‘Up until then, no one had ever looked at or cared about the properties of an individual molecule, they cared about an ensemble,’ says Steven Lee, a former postdoctoral researcher in Moerner’s group at Stanford University, US, who is now based at the University of Cambridge in the UK.
The next big step was the jump to room temperature
W E Moerner
At the time, Moerner wasn’t thinking of microscopy or cell biology, but working at IBM’s Almaden Research Center in San Jose, US, exploring ways to store information optically. He had no idea that his measurements would go on to spark a revolution in microscopy. ‘But I knew we would see surprises, because we simply just hadn’t measured this before,’ says Moerner. He had to carry out his single molecule detection experiments at temperatures of just 4K. ‘The next sort of big step was the jump to room temperature,’ he says.
This is something Betzig – working at AT&T Bell Laboratories in Murray Hill, US – achieved in the mid-1990s, using a technique called scanning near-field microscopy to image a single molecule of carbocyanine dye at room temperature.
He also began to think about how single molecule detection might be used to achieve super resolution. At this point, single molecules could only be detected at room temperature when diluted to nanomolar level, and spread far enough apart to prevent the fluorescent signals they emitted overlapping. This was clearly no good for imaging the molecules in cells that exist in much higher concentrations.
But Betzig thought that if differently coloured fluorescent molecules can be used to label the sample – each spread through the sample at low density – the microscope could locate all the molecules of each colour in turn and the images could be superimposed to give a complete picture.
Betzig published his theory in 1995,4 but at the time there wasn’t a way to put it into practice, because molecules with the right optical properties did not exist. He then decided to take a break from research, and resigned from Bell Labs to work as vice-president of R&D at Ann Arbor Machine Company, his father’s machine tool business.
Palm reading
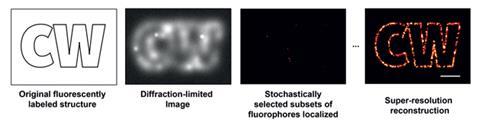
Meanwhile, single molecule spectroscopy continued to grow as a discipline. Two years after Betzig published his ideas, Moerner – then based at the University of California in San Diego, US – made a hugely important yet somewhat accidental discovery. Working with a molecule of green fluorescent protein (GFP, itself a discovery that was awarded the chemistry Nobel in 2008; see A glowing green Nobel) the group found GFP ‘blinked’ when excited with a laser beam in the single molecule microscope. The protein went through several cycles of fluorescence and darkness before eventually fading. Furthermore, the fluorescence could be turned on and off by altering the beam – irradiating dark molecules at 405nm could re-activate them. ‘We just didn’t expect at room temperature to see such interesting photodynamics,’ says Moerner. ‘That was very exciting.’
This ability to turn fluorescent molecules on and off opened up the possibility of distinguishing between individual molecules in a concentrated medium like a cell – the molecules would not have to be separated spatially or by fluorescing with different colours – they could be activated at different times instead. Seeing the work of Moerner and others with GFP was enough to tempt Betzig back into the field. He left the family company, and together with friend Harald Hess – a former colleague from Bell Labs who was also unemployed – developed photoactivated localisation microscopy (Palm), building the first prototype device in Hess’s apartment in San Diego.
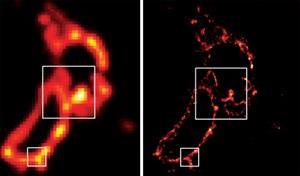
They were helped by cell biologist Jennifer Lippincott-Schwartz at the US National Institutes of Health in Bethesda, who had developed a mutant GFP that initially existed in the ‘off’ state, but could be made to fluoresce by irradiating it at 488nm, which eventually caused it to darken irreversibly, a phenomenon known as ‘photobleaching’. This allowed a widely dispersed set of molecules in a sample to be activated, located and imaged, then silenced, a cycle that can be repeated over and over again to build up a complete picture of all the molecules and reveal an image at a resolution far higher than Abbe’s limit. The first demonstration of the Palm technique came in 2006, when it was used to image whole cells and subcellular structures including mitochondria tagged with fluorescent proteins.5
The same year, another US group led by Xiaowei Zhuang at Harvard University developed a variation on the technique called stochastic optical reconstruction microscopy (Storm) using fluorescent cyanine dyes that can be reversibly switched on and off.6
Beyond biology
Today, methods like Sted, Palm and Storm are offering cell and molecular biologists a nanoscale view inside cells without the constraints of electron microscopy. Lippincott-Schwartz says the techniques are ‘revolutionising our concepts of some of the most fundamental concepts of living cells, which until now were hidden’. Her group at NIH have continued to use super-resolution imaging to investigate how proteins organise themselves on different cellular structures, including those of disease-causing bacteria and viruses. ‘The techniques could even be used in identifying unique features of the Ebola virus to help find new ways to abolish it,’ she suggests.
Moerner has also collaborated with colleagues at his current base at Stanford University, US, to examine the contents of cells. His group and Lucy Shapiro’s have used single molecule spectroscopy to study the organisation of proteins involved in cell division in bacteria, and he also worked with Judith Frydman’s lab to look at the aggregation of proteins involved in Huntingdon’s disease, an approach which may offer new targets for treatments.
Michel Orrit, another early pioneer of single molecule spectroscopy now based at Leiden University in the Netherlands, points out that the field also has a lot to offer other areas of research. ‘This Nobel prize shows only one aspect: the imaging,’ he says. His group, and many others, are applying single molecule spectroscopy to areas such as biophysics, quantum optics and materials science. ‘Our group, for example, has been studying glass forming, and how viscous liquids dissolve,’ he says. ‘You can also start to understand catalysis on a much more detailed basis or how molecules diffuse in nanotubes. And in this way you become more aware of the quality of your material.’
And as the techniques become faster, less expensive and more user-friendly, more and more people are beginning to use them. ‘It’s really widespread. If you speak to most people they will have heard of super-resolution imaging no matter what their background,’ Lee says.
‘They can use the instruments now, the code becomes more automated and it’s much more robust than it used to be,’ he adds. ‘It’s opening the door to a lot of fundamental questions that couldn’t really be asked before.’
For a field that is advancing so rapidly, and is closely tied to improvements in the technology as well as increasing numbers of users, it is difficult to see what will come next.
‘We have a wide playing field,’ says Hell. ‘[And there are] different types of optical microscopes that all have very different characteristics – some are very fast, others are very sensitive, others are very accurate. This will improve even more in the future.’
Those developing the tools continue to push for higher and higher resolution. ‘It’s a really exciting area to be in,’ says Lee. ‘The resolution of localisation based tools [such as Palm and Storm] is limited by the number of photons you get out of a fluorophore – at the moment, we’re finding we’re going to have to develop better dyes to be able to give us more photons.’
Continuing improvements to the capabilities of super resolution imaging will allow scientists to tackle questions of ever increasing complexity, says Moerner. ‘There are so many interesting systems to study. We are going to continue to push the frontiers everywhere we can.’
References
1 T A Klar et al, Proc. Natl Acad. Sci. USA, 2000, 97, 8206 (DOI: 10.1073/pnas.97.15.8206)
2 F Görlitz et al, Prog. Electromagn.Res., 2014, 147, 57 (http://rsc.li/1vmZ7Ss)
3 W E Moerner and L Kador, Phys. Rev. Lett., 1989, 62, 2535 (DOI: 10.1103/physrevlett.62.2535)
4 E Betzig, Opt. Lett., 1995, 20, 237 (DOI: 10.1364/ol.20.000237)
5 E Betziget al, Science, 2006, 313, 1642 (DOI: 10.1126/science.1127344)
6 M J Rust, M Bates and X Zhuang, Nat. Methods, 2006, 3, 793 (DOI: 10.1038/nmeth929)









No comments yet