For the first time, researchers have observed a mysterious phenomenon known as the chirality-induced spin selectivity (Ciss) effect in isolated molecules – unattached to substrates or solid electrodes. ‘[This] experiment verifies that the [Ciss] effect results only from the system’s chirality,’ says Ron Naaman from the Weizmann Institute in Israel, who discovered the Ciss effect in the late nineties. Naaman, who was not involved in the new work, says that the findings ‘reveal the mechanism of the Ciss effect’ and could have direct applications in quantum computing, energy generation and efficient lighting.
‘When we measure the direction of spin relative to an external magnetic field, it can have only two values, either with or against the field,’ explains Michael Wasielewski from Northwestern University, US, who led the project. ‘If electrons move through a chiral material […] only one of the spin directions is transmitted, while the other is inhibited,’ he adds. ‘If the material has the opposite handedness, the effect is reversed.’ This effect, known as Ciss, establishes a strong link between spin and charge motion and therefore has strong implications in macroscopic properties. ‘It could have important implications for biology, as well as materials for quantum information and energy,’ adds Wasielewski.
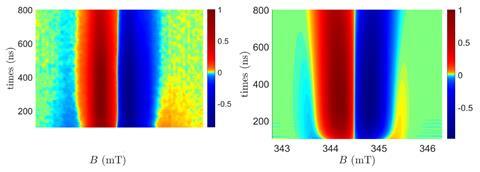
Wasielewski’s team synthesised molecules that contain three components: a photoresponsive electron donor, a chiral bridge, and finally an electron acceptor. ‘A short laser pulse excited the donor … and as a result an electron was transferred from the donor, through the bridge to the acceptor,’ explains Naaman. The electron transfer was monitored using time-resolved electron paramagnetic resonance spectroscopy (EPR), which yielded spectra for each of the enantiomers of the bridge.
These measurements are particularly hard because researchers must generate a charge-separated excited state of the molecule in which the electron transfer is ‘slow enough’ to [measure] by EPR, explains Jeanne Crassous, an expert in chiral systems based at the University of Rennes in France. ‘[It was] a good idea to take advantage of a liquid crystal to fix the orientation of the chiral molecule at low temperature,’ she adds. ‘By orienting [each] enantiomer in a liquid crystal and applying a magnetic field in two opposite directions, they found different shoulders in the shape of the EPR.’ However, in control experiments featuring molecules with an achiral bridge, no change was observed. ‘[This] demonstrates that the electron transfer depends on the chirality of the molecule.’
Until now, ‘all examples of [the Ciss] effect essentially involved attaching chiral molecules to metals or semiconductor surfaces,’ says Stefano Carretta, from the University of Parma, in Italy, who co-led the study. ‘We directly demonstrate for the first time that Ciss is an intrinsic property of electron motion within the molecules themselves, independent of the involvement of interfaces or the attachment to substrates,’ he adds. ‘This is crucial to understanding the phenomenon and implementing possible applications.’
Because electron spins in magnetic molecules could find applications as qubits, Ciss could be exploited in quantum computing applications. ‘Interestingly, Ciss is very efficient even at room temperature, opening the possibility to increase the operating temperature of molecular qubits,’ says Carretta.
Other applications include organic light-emitting diodes (OLEDs) and biocatalysis. ‘Ciss based OLEDs emit circular polarised light, … [which] may allow the production of much more efficient devices,’ explains Naaman. ‘Ciss [could] explain why chirality was conserved so persistently throughout evolution, [present] in all forms of life,’ he adds.
Crassous also believes that Ciss effects could underpin some important aspects of biology. ‘Nature has chosen chiral molecules to perform some biological functions really efficiently,’ she says, noting that Ciss effects could explain the efficiency of enzymatic and biological processes that involve electron transfers. ‘There’s still a lot to discover and understand!’
References
H J Eckvahl et al, Science, 2023, 382, 6667, DOI: 10.1126/science.adj5328
















No comments yet