Using a photocatalyst to move ortho- and para-substituents one atom along a pyridine ring could simplify how chemists make meta-substituted aromatic molecules, a key scaffold for several drug compounds.
Adding electrophiles at the meta position of a substituted aromatic ring is challenging, as these reactions often go through a less stable intermediate, compared with the same addition at the ortho and para positions. Electron-withdrawing nitro or acyl groups, for example, can make the meta position more favourable, but these groups are often unwanted and slow down reactions. Meta-substituted compounds are common in pharmaceuticals, such as the cancer drug imatinib, though scientists are looking for more efficient ways to synthesise such compounds without needing directing groups or palladium cross-coupling reactions.
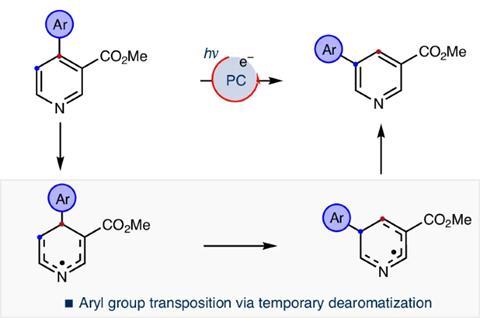
Researchers have developed a way to move substituents along the ring to the meta position, by exposing pyridines arylated at the ortho or para position to visible light in the presence of an organic photocatalyst.
Computational analysis revealed that the aryl substituent gains electrons from the catalyst and then forms a cyclopropane ring with the pyridine. The initial carbon–carbon bond then breaks, shifting the aryl substituent to the meta position.
The team was able to use this strategy to move a variety of aryl substituents in nicotinates – pyridines with a methyl ester. In pyridines with two different aryl substituents, the team found that various factors – including electronics, sterics and the relative position of the groups – influenced which aryl group moved.
The team hopes that the method can help simplify synthetic routes to molecules with meta substituents and is looking to apply the strategy to other heteroarenes.
References
E Y K Tan et al, Nat. Synth., 2026, DOI: 10.1038/s44160-026-01023-6















No comments yet