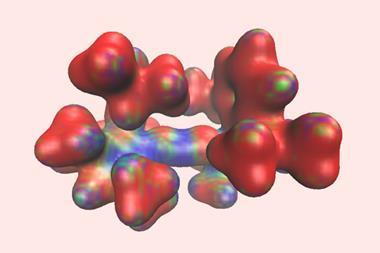
Researchers in Canada have created an N-heterocyclic carbene ligand with the highest buried volume reported to date.1 The exceptionally sterically encumbered ligand features N-bound trityl groups and a benzylated backbone, and its steric and electronic nature allows it to access low‑coordinate gallium and lithium complexes.
Bulky ligands can enforce rare low coordination in inorganic elements, often giving rise to unusual properties that are exploited in catalysis. Among the most versatile of these ligands are N‑heterocyclic carbenes (NHCs), which feature a neutral carbon atom bearing two unshared electrons, stabilised by two ring nitrogen atoms. Their stability, capacity to form strong metal–carbon bonds and tuneable steric and electronic characteristics have made NHCs a versatile and widely used class of ligands.
In 2017, a team lead by Eric Rivard from Alberta University reported the extremely bulky NHC ITr (ITr = [(HCNCPh3)2C:]), which featured two nitrogen-bound sterically shielding trityl (CPh3) substituents.2 At the time, it represented the highest percent buried volume (%Vbur) reported to date for an NHC and allowed access to a thermally stable quasi-monocoordinate thallium(i) cation complex.
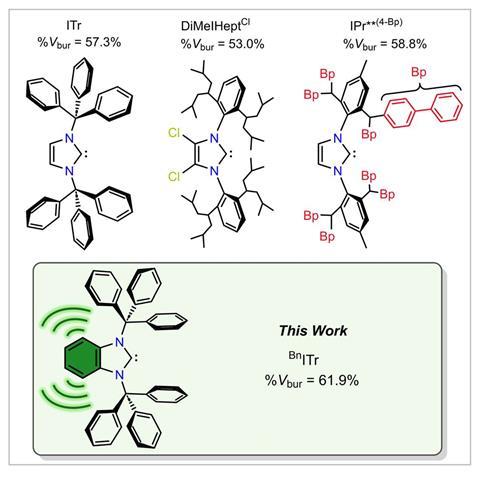
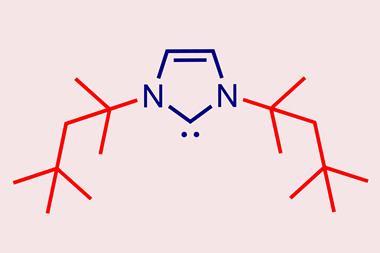
Now, Rivard and Ludwig Zapf have made a benzylated analogue, NHC ligand BnITr (BnITr = [(C6H4){NCPh3}2C:]). Using a two‑step synthesis from commercially available 2‑trityl‑benzimidazole, they produced a ligand with a %Vbur exceeding 60%. This surpasses the previous record of 58.8% set by an NHC ligand with biphenyl wingtips reported by Michal Szostak’s group at Rutgers University, US, in 2025.3
Single-crystal x-ray diffraction showed that the benzylated backbone in Rivard and Zapf’s ligand forces the trityl groups to rotate towards the carbene moiety. Such rotation increases the steric bulk beyond that of the non-benzylated ITr, resulting in a highly shielded carbene centre.
The duo used this new bulkier carbene to stabilise a quasi-monocoordinated gallium(i) cation complex and isolate a previously elusive quasi-monocoordinated lithium cation complex. The lithium-based complex represents the first example of a monomeric molecular complex without any extra ligands or solvents at the lithium centre. Zapf and Rivard also attempted to exploit this ligand for the synthesis of a quasi-monocoordinated palladium(0) complex, an intermediate believed to be key in palladium-catalysed cross-couplings. Surprisingly, the synthesis did not yield the expected one-coordinated [BnITr–Pd] complex but instead a compound where one trityl group migrated to the palladium centre.
References
1 L Zapf and E Rivard, Chem. Sci., 2026, DOI: 10.1039/d5sc09514j
2 M M D Roy et al, Chem. Eur. J., 2017, 23, 11249 (DOI: 10.1002/chem.201703215)
3 Y Sha et al, Organometallics, 2025, 44, 1848 (DOI: 10.1021/acs.organomet.5c00232)


















No comments yet