Ligand selection unleashes first monomeric [Cu(η6-C6H6)]+ complex
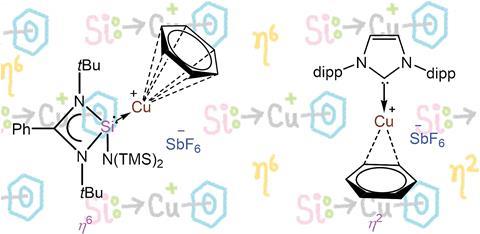
Inorganic chemists working in Spain and India have isolated the first unsupported example of a monomeric copper cation where the copper atom bonds to a benzene ring in the η6 bonding mode.
All previous research has suggested that the η1/ η2 bonding modes are overwhelmingly preferred due to repulsion between the central metal d-orbitals and the benzene ring. While tethered examples of the η6 mode were known, the ability to isolate a monomeric [Cu(η6-C6H6)]+ complex was thought to be highly questionable.
Inspired by recent work on silylene–copper bromide complex, [{PhC(NtBu)2 }Si{N(SiMe3)2 }]2Cu2Br2, a team led by Santiago Alverez, Jorge Echeverría and Shabana Khan at the University of Barcelona and IISER Pune, used increased back donation from the neutral ligand to access [{PhC(NtBu)2SiN(SiMe3)2 }Cu(η6 - C6H6 )]+[SbF6 ]–.
Through further experimental study with other ligands and DFT, they found that to favour the η6 mode, the co-ordinating ligands had to have π-acceptor characteristics, with a third row donor atom – to minimise steric repulsions – and a small cone angle.









No comments yet