A new electrochemical method can turn strained rings into versatile platforms for carrying out multiple precise transformations in a single sequence.
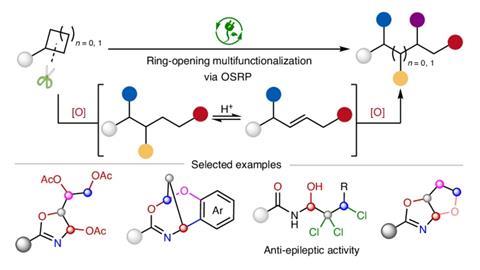
Chemists have long prized strained rings as spring-loaded starting points for building complexity. However, once the ring is opened, the molecule quickly loses reactivity, and synthesis usually stops after adding two groups across a single bond. Useful as this is, it barely taps the full potential of these high-energy structures.
The new advance hinges on an unexpected idea. Tao Shen of Shanghai Jiao Tong University says their pool of ‘slow-release olefins’, which serve as controllable intermediates for the reaction, acts like a pressure valve. ‘Instead of unleashing all the reactive potential at once, our … system continuously generates reactive olefin intermediates at low concentrations in a slow, controlled manner,’ he says.
The reaction relies on two key additions: strong acids that reversibly generate olefin intermediates and electrochemical oxidation, which lets the chemists control the reaction rate.
‘Following initial ring-opening, [the acids] generate [a pool of] reactive olefin intermediates,’ Shen says. ‘These intermediates undergo electrochemical oxidation, enabling sequential installation of additional functional groups. By iterating this “release-and-activate” cycle, we achieve remarkable one-pot functionalisation of up to four sites.’
The result is an unusual level of control over where and how bonds are transformed, even across typically inert C–H and C–C sites. This strategy also prevents runaway reactions like polymerisation while keeping the molecule reactive enough for further modification.
‘This reaction is mind boggling as I would not have predicted such polyfunctionalised products from simple cyclopropane derived substrates,’ comments Song Lin at Cornell University, who was not involved in the study. ‘It shows how electrochemistry can activate molecules in unconventional ways and enable access to intermediates and products that are otherwise unimaginable.’
By tuning electrochemical conditions, including direct current, rapid alternating polarity and electrophotocatalysis, the team could also direct reactivity to more remote positions, enabling alkenylation far from the original ring. The products are not just highly functionalised curiosities, but they include synthetically useful motifs such as oxazolines, polyols and polyhalogenated alcohols, along with more complex bicyclic frameworks formed through skeletal rearrangement.
‘Hydroxy-trihaloamides serve as potential building blocks for antiepileptic drugs,’ says Shen. ‘The unique combination of chlorine/bromine atoms with a hydroxyl group in these compounds presents significant synthetic challenges via conventional methods.’
Beyond expanding the toolbox for strained-ring chemistry, the work points to a broader shift: electrochemistry as a means of choreographing stepwise reactivity in situ, rather than forcing all transformations to occur at once. If generalisable, this ‘slow-release reactivity’ concept could offer chemists a new way to tackle one of synthesis’s toughest challenges: selectively transforming multiple inert bonds within a single molecule.
References
Y Li et al, Nat. Chem., 2026, 18, 656 (DOI: 10.1038/s41557-026-02110-z)














No comments yet