Using electrons instead of reagents offers many potential benefits, but there are still barriers to overcome, as Rachel Brazil reports
Industry’s reliance on petrochemicals is one of the main reasons why electrochemical synthesis was never fully explored says Tobias Gärtner, chief executive at ESy-Labs, a start-up located in Regensburg, Germany, that specialises in electrosynthesis technology. Modern industrial organic chemistry has evolved to efficiently exploit fossil fuel-based hydrocarbon feedstocks and turn them into chemical products using classical organic chemistry, from the nylon fibres in our clothes to the artificial flavours in our foods.
The carbon toll of these industries – the chemical sector being the third largest industrial emitter of carbon dioxide – and a legacy of polluting waste is leading chemists to search for greener processes. And they are turning to electrosynthesis: using an electric current to facilitate chemical reactions instead of chemical redox agents. ‘Electrochemistry was a niche [method] but more and more it’s coming out of the niche and being recognised as a real synthetic method,’ says Gärtner. But while publications and funding in electrosynthesis have been on the rise in the last decade, academic trends don’t always successfully make their way to industry.
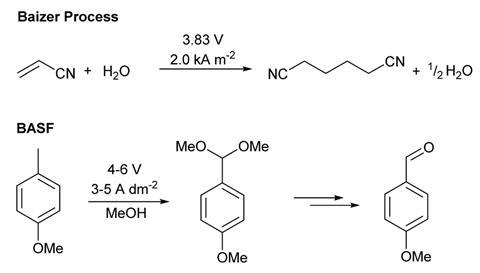
Electrosynthesis has never been absent from the chemical industry. Interest tended to rise in times when crude oil prices rose or electricity prices fell, points out one of ESy-Labs’ co-founders, Siegfried Waldvogel from Johannes Gutenberg University Mainz in Germany who has been working in electrosynthesis for 30 years. One of the earliest examples from 1849 is the Kolbe reaction, the electrosynthetic radical coupling of two carboxylic acids. There was also an upsurge in interest in the 1960s with the Baizer process developed by Monsanto. This cathodic reduction of acrylonitrile to adiponitrile is used to annually produce in the range of 100,000 tonnes of the polyamide nylon-6,6, a superior form of nylon, made from hexamethylenediamine and adipic acid (hexanedioic acid).
The latest resurgence comes with the challenge to decarbonise the chemical industry and the hope that cheaper renewable electricity can be used to fuel these reactions. This is certainly the case for biotechnology company Vertex Pharmaceuticals, who focus on rational design approaches to drug discovery. ‘The ability to do away with reagents and just use electricity, especially if it comes from a green source, is certainly a consideration,’ says Vertex principal scientist Robert Green.
Looking for shortcuts
Agrochemicals specialist Syngenta started looking at electrosynthesis around 2017, after Waldvogel gave a talk at their research labs in Switzerland. Chris Scarborough, who was then working in process chemistry, says he was particularly struck by the tendency in industry to avoid direct oxidation reactions which are often dangerous, and instead use workarounds involving far more steps including nitrations, reductions or diazotisations. Electrosynthesis could offer more direct routes, plus a simple safety lever. ‘If there was a problem, cutting the electrical supply could also stop a runaway reaction or something dangerous happening,’ says Scarborough.
The other safety advantage is the removal of toxic reagents currently used in many conventional organic syntheses, including noble metal catalysts. ‘[This is important] especially in the pharmaceutical industry where you have to be sure that there is no contamination,’ says Waldvogel. Electrosynthesis also promises less waste. ‘[For example,] if you’re not using sodium borohydride as a reducing agent and producing boron oxides as byproducts, it’s potentially a much cleaner synthetic approach,’ says David Hodgson, a specialist in industrial electrochemisty and chief technology officer at advanced materials producers Technical Fibre Products.
Cost at scale is the bottom line for most industry reactions, although that isn’t always the case for medicinal chemists, because the value of an active pharmaceutical ingredient is so high compared to bulk or even fine chemical, says Pierre-Georges Echeverria, R&D director at US sustainable specialty chemical company Pennakem. ‘For medicinal chemists [they are looking for] short cuts in the synthesis,’ he adds. ‘When the chemistry is straightforward, even if the yield is not that good, they don’t care: they have the molecule and that’s great.’
The hope is also that electrosynthesis may provide access to new chemistries via the free radical intermediate species that are produced in an electrolysis cell. ‘The chemical reaction concept behind [electrosynthesis] is in most cases completely different compared to conventional chemical reactions,’ explains Gärtner. ‘There are loads of examples of making interesting heterocycles that were quite frankly a pain to make, that you can [make more easily] and pharma and agrochemicals are stuffed full of interesting heterocycles.’
Size matters
The problem for industry is always scaling up the reactions developed in academic labs. Echeverria says he started experimenting with electrosynthetic oxidations of secondary alcohols in 2016 at Minakem, a sister company focused on making active pharmaceutical ingredients. He was trying to reduce the oxidant waste generated. ‘It works pretty well, [but] at that time we gave up on this topic, due to the lack of scale up solutions.’ A lack of standardised equipment for scale is still a limitation facing industry, he says.

The team at Syngenta has also grappled with scale, and particularly moving between the different scales they need for fast early exploration and then moving to producing larger amounts, all ideally running under the same conditions. Process research chemist Matthias Lehmann says they now have two systems at the 100mg scale to be able ‘to deliver an answer to whether a transformation is possible or not’. But they found that although commercial equipment existed for very large scale manufacturing there was nothing to evaluate industrially relevant scale-up conditions at the gram scale, so they designed their own kit, which they still use today.
When scaling up, ‘typically all these processes switch to flow’, says Green. Flow chemistry allows reactions to run in a continuous stream rather than in batches and is a well-established technique for large scale manufacturing. At scale, working in flow is crucial because in batch the size of the necessary electrode surface would also need to be scaled up, making the whole cell unmanageably large.
Can we do an electrosynthetic reaction here, and will this save steps or waste?
The problems of mass transport of reactants at larger scales is even greater for electrosynthesis than for conventional scale ups. ‘You need to transport starting materials to your two electrodes and remove the product from your electrodes and this has to be matched with the reaction kinetics,’ explains Syngenta research chemistry team leader Andrei Iosub. Syngenta have experimented with adding mixers to their electrochemical flow cell to increase mass transfer rates.
The number of companies who are introducing new electrosyntheses is not clear says chemist Kevin Lam from the University of Greenwich in the UK. He has worked with both GSK and AstraZeneca, but he says companies are not always open about their new strategies so it’s difficult to know. Some companies never stopped electrosyntheses; Lam recently noticed German chemical company BASF have long-standing patents on electrosyntheses that have only just been published in the academic literature.
Syngenta are so far only working at a small scale. ‘Whenever we have an interesting oxidative transformation, we think “OK, can we do an electrosynthetic reaction here, and will this save steps or waste?”’ says Iosub. They are also thinking carefully about how some of the traditional ingrained workarounds to avoid direct oxidations could provide opportunities for simpler direct electrosynthesis. ‘We found that there are plenty of exciting opportunities for electrochemistry in our environment,’ says Scarborough. The team are sure it’s just a matter of time before one such reaction is scaled up for production.
Vertex are also in the early stages. ‘We’ve developed some internal capabilities to be able to quickly optimise and screen different reactions’ but, says Green, ‘we’re still assessing the literature, understanding where the best impacts can be made.’
Electrode design
Since his first foray in electrosynthesis, Echeverria and colleagues partnered with leading academic electrosynthetic chemist, Phil Baran from the Scripps Research Institute in California, US, to see if they could develop greener and more cost-effective electrosyntheses. They developed a furan oxidation to synthesise 2,5-dimethoxydihydrofuran (DMDHF), used to make a wide variety of valuable chemicals such as pyridazine used in agrochemicals and flavour enhancer maltol. The furan starting material came from the bio-based sugar dehydration product furfural (C₄H₃OCHO) and the pilot was able to produce a kilogram per week, getting to a current efficiency of 88% (meaning 88% of electrons delivered contributed to the desired reaction). Echeverria says this process would compete with DMDHF produced conventionally in China and could be ‘a game changer’.
The Minakem team also worked with Baran to develop a carbonyl desaturation reaction that could proceed without the large amounts of expensive palladium catalyst used in the conventional synthesis. This type of reaction adds a carbon–carbon double bond next to a carbonyl group to open up downstream reactivity. They came up with a simple process, scalable to 100g.
The electrode material and design is one of the crucial factors in electrosyntheses. ‘If you don’t get the right electrode material, the right current density and the right engineering, you can end up making a very different mix of products than you would want, so that affects selectivity and it affects yield,’ says Hodgson. ‘Things like [electrode] porosity is going to be important.’ His company are looking at the type of advanced materials that might improve electrode performance.
It’s 200% efficiency if both electrodes are productive
Optimising the factors contributing to successful electrosyntheses, particularly the choice of electrode, is often trial and error. ‘If you have a lot of experience, you have a kind of intuition, of course, but the problem is, even in my case, I was sure that in [a particular] reaction, this electrode should perform much better but the experimentation turned out differently,’ says Waldvogel. His start-up ESy-Labs, founded in 2018, is hoping to change that.
‘When you switch from 25 to 35°C, you see a dependence on the reaction, but switching from copper to carbon and there is no dependence,’ explains Gärtner. ESy Labs is using AI and other statistical methods to better optimise processes. They are carrying out high throughput electrosyntheses – from 40–80 reactions in parallel – to create enough data points to train an AI system to identify the best electrode material, solvent or electrolyte for any given reaction and hope this will aide in designing new processes.
Lam says that the holy grail for industrial electrosynthesis would be a paired electrosynthesis where a useful product is produced at both the anode and cathode. ‘That’s really fantastic – it’s 200% efficiency because both electrodes are productive.’ A long established example is German chemical manufacturer BASFs production of the aromatic aldehyde lysmeral (butylphenyl methylpropional) which provides an artificial lily of the valley scent, once produced at the 10,000 ton per year scale (it is now banned from cosmetics in the EU and UK due to its endocrine disrupting properties). The electrosynthesis produces an intermediate methoxy benzaldehyde at the anode which undergoes further reactions to form lysmeral. At the cathode a benzenedicarboxylic acid is reduced forming phthalide, a chemical used to produce fungicides. ‘It’s what I would consider the slam dunk application, if you can find one and scale up the electrochemistry,’ says Iosub
A hybrid approach
Jean-Philippe Tessonnier, a chemical and biological engineer at Iowa State University in the US, is trying another approach: hybrid microbial electrosynthesis. This combines the power of biocatalysis and the advantages of electrosynthesis, using biomass feedstocks. With low-cost renewable electricity this starts to look economically attractive.
Tessonnier says that biosyntheses using bacteria or yeast can be efficient at some chemical conversions but not others – particularly removing carbon–carbon double bonds. ‘Maybe biology should focus on what it does well, and let chemistry do the rest,’ he explains. The method developed with colleagues at the Center for Biorenewable Chemicals initially aimed to produce adipic acid – the nylon feedstock usually derived from petrochemicals through multiple oxidation steps. Over 3 million tons of it are made annually, producing a similar amount of nitrous oxide greenhouse gas.
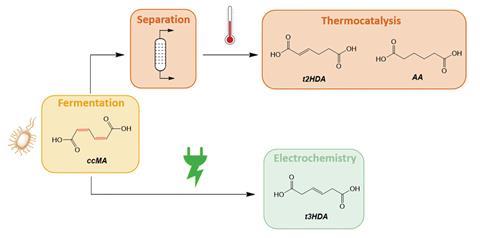
Rather than separating the phenol starting material produced in the fermentation broth from other impurities, Tessonnier decided to see what would happen if he just stuck in some electrodes. ‘Fermentation broths contain a lot of salts, magnesium sulfate and other things, so this already looks like an electrolyte,’ he reasoned. In 2021, their first experiments, microbially converted sugars or lignin monomers into the dicarboxylic acid cis,cis-muconic acid (C6H6O4), which they were then able to electrochemically hydrogenate to remove the double bond and form trans-3-hexenedioic acid at very high yields. While not their intended product, it is also a valuable monomer because it can be used to produce nylon 6,6 with attached functional groups to introduce novel properties.
Tessonier has now also published a hybrid method to produce adipic acid, using supported palladium nanoparticles on carbon as a catalyst which facilitates electron transfer and the subsequent reduction to adipic acid on surface terrace sites. Other groups are pursuing similar approaches including a Kolbe electrosynthesis to couple medium chain fatty acids from fermented biowaste to produce hydrocarbon fuels.
Although electrosynthesis undoubtably has the potential to be greener, it may not always be the best solution according to Lam. The electricity costs are not negligible: ‘At large scale, every single volt will consume more money, so you need to have a very efficient process,’ and he says there will still be waste. ‘We have to add a large amount of supporting electrolyte – not always, but very often for reactions, and it doesn’t contribute to anything in the reaction.’
‘What I haven’t seen much of in electrochemistry yet is really complex molecules,’ concedes Green. ‘Big molecules that have got multiple functionality.’ Through controlling the potential, you can ‘tune in’ the reactivity to a specific part of a molecule but ‘Fundamentally, you can only tune in to the lowest energy [reaction] – that’s always going to go first,’ he says. It’s also difficult to control stereochemistry. For now it is largely restricted to producing the earlier building block molecules, but clever catalysts or electrode design could provide further control and Green hopes the research community will come up with solutions. ‘As people apply it to more complex molecules, we’ll see how far can you push it.’
Electrosynthesis is not magic
In the meantime, moving electrosynthesis into industry suffers from the same problems as the adoption of any new technology. ‘The day to day pull back towards the normal chemistry can be so strong that you can lose focus,’ says Scarborough, But he says the team at Syngenta aren’t giving up.
More unique to this technology is the mismatch in the skills set of many synthetic organic chemists. Lam jokes that many of them were likely traumatised by physical electrochemistry as undergraduates. Scarborough has seen similar reservations. ‘When I started [doing electrosynthesis] at Syngenta, I had some people looking at me like I was crazy to plug this reaction into the wall,’ he remembers. ‘People were very nervous about the set up originally’.
‘Companies are packed with people who have been using thermal chemistry and thermal catalysis for decades, so it takes time to educate people and make them just willing to listen to you and look at potential benefits,’ says Tessonnnier. In his experience pharma companies seem most open to change.
But Waldvogel is convinced electrosynthesis will have a huge impact. ‘I’m very confident that it’s not a bubble… just keep in mind, you can be more oxidising than fluorine gas and more reducing than caesium…. a lot of things are possible.’
The ultimate challenge is to activate carbon dioxide electrochemically to use as a synthetic building block, an area Lam is working on. But he concludes ‘electrosynthesis is not magic’, and it can sometimes be oversold. It’s not going to replace conventional chemistry. ‘These are different technologies and complementary technologies,’ says Tessonnnier . But it does offer an alternative way to drive a chemical reaction and another tool in the industrial chemist’s toolbox.
Rachel Brazil is a science writer based in London, UK














No comments yet