
An elusive nitrene-like arsenic intermediate has been isolated and characterised for the first time. Analysis has confirmed many of the properties predicted for arsinidenes, but the unusual reactivity suggests heavier main group elements may have more unique chemistry than previously thought.
Carbenes and nitrenes are powerful synthetic intermediates, each containing a neutral centre with two unpaired valence electrons. Although highly reactive and unstable, the versatility of these reagents has made them a cornerstone of synthetic chemistry and both their fundamental nature and their wider reactivity have been extensively studied. However, far less is known about heavier related compounds like arsinidenes, which are too short-lived to characterise. ‘With electron deficient species you need to have substituents to stabilise the system,’ explains Artur Mardyukov, a physical organic chemist at Justus Liebig University in Germany. ‘In the case of carbenes and nitrenes, the orbital overlaps are efficient. But for arsenic the orbitals are more diffuse and the overlap is poor so they’re difficult to stabilise.’
Mardyukov’s team used a technique called matrix isolation, freezing the in situ generated arsinidene in solid argon to extend its lifetime. Temperature-controlled photolysis of a diazide precursor then liberated the highly reactive phenylarsinidene, which was immediately condensed in argon at 10K. The inert solid surroundings prevented any further reaction, enabling the team to thoroughly characterise the individual molecules trapped within the matrix. Spectroscopic analysis revealed a C–As bond length of 1.932Å – compared with 1.318Å for the analogous nitrene C–N bond – and confirmed the suspected lack of stabilising delocalisation over the phenyl substituent. ‘The precision characterisation is good and confirms the properties we would expect,’ comments Rory Waterman, an inorganic chemist at the University of Vermont, US. ‘ But what I found particularly interesting was the divergence in reactivity with oxygen between the arsinidene and its lighter congeners which they observed subsequently.’
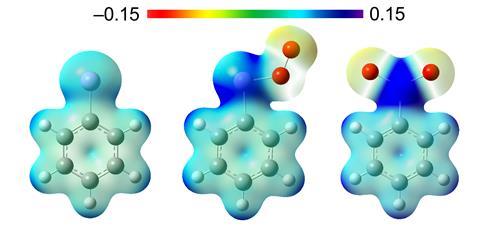
The team probed the reactivity of the matrix-bound phenylarsinidene with molecular oxygen, a known reaction with both phenylnitrene and phenylphosphinidene. Whilst in each case, the reaction gives the dioxo product, spectroscopic experiments supported by computation revealed some unexpected mechanistic differences. Each pnictogen species underwent a different initial interaction with the oxygen, either reacting via a different mode or through a different conformation, and surprisingly, the arsinidene behaved more like the nitrene than the phosphinidene. ‘We computed the potential energy surface to understand why these analogous species interact differently,’ explains Mardyukov. ‘Which conformers or types of intermediates are more stable will affect whether they react through kinetic or thermodynamic control. This stability depends on if you have nitrogen, phosphorus or arsenic and can be explained from molecular orbital theory.’
‘The mechanistic differences demonstrated here between the low-valent pnictogens encourage further study. It is those differences that may identify new reactivity of greater practical interest,’ comments Waterman. ‘Overall this work provides a better understanding of arsinidene, which allows us to access and use it more effectively and will motivate more chemists to find the unique reactivity of arsenic in flasks.’
Mardyukov’s team are continuing to investigate the potential of these underutilised intermediates, both at a fundamental level and as a tool for synthesis. ‘The synthesis of arsinidene is general, mild, and surprisingly simple,’ he says. ‘I hope ultimately people will use it to incorporate arsenic into their chemistry.’
References
W Qian et al, J. Am. Chem. Soc., 2023, DOI: 10.1021/jacs.3c02935














No comments yet