By playing tug-of war with individual molecules, Chinese scientists have shown that mechanical force can solve a stability problem facing drug antibody conjugates in large scale. Wei Wang and Yi Cao from Nanjing University and colleagues first stabilised maleimide–thiol linkages by pulling on them in intricate atomic force microscopy (AFM) experiments. They then used force from ultrasonic energy to stabilise the linkages, which connect conventional small molecule drugs with antibodies in cancer therapies like ado-trastuzumab emtansine (Kadcyla).
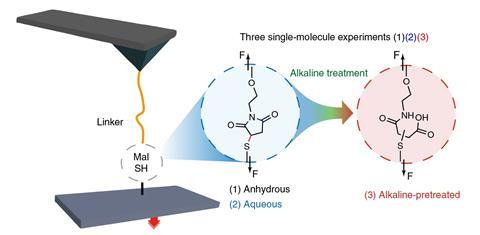
The sulfur atom in maleimide–thiol linkages can be a key weakness, as sulfur atoms in other molecules can displace it, or it can spontaneously detach in a retro-Michael reaction. The Nanjing team therefore saw the bond between the thiol group and the maleimide ring as reversible, Cao tells Chemistry World.
The scientists investigated the link using an AFM probe with a cantilever with a maleimide molecule attached to it. They pushed the probe against thiol molecules attached to glass plates carrying thiol molecules, forming a linkage in the forward Michael reaction, then pulled them apart. Repeating the process hundreds of times, the Nanjing researchers discovered that the linkages did not break as easily as they expected. ‘Surprisingly we found that the maleimide–thiol linkages were not reversible in water,’ Cao explains.
The team varied the conditions, recording forces indicating that the Michael reaction was exactly reversible when water was not present. In water, they sometimes observed a retro-Michael reaction and sometimes another process that required more force, which they reasoned was the maleimide’s ring structure springing open. When they made the watery conditions alkaline to open the ring before their experiments, they only observed high rupture forces.
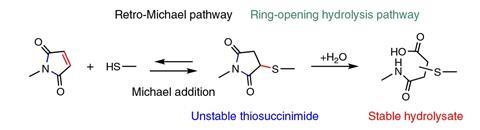
In these experiments, mechanical force enables reaction pathways that are not otherwise possible, Cao explains, because it changes activation energy barriers. ‘The rate constant of the reaction is exponentially dependent on the mechanical force,’ he adds.
Finally, Cao and his colleagues opened maleimide rings, producing more stable linkages in conjugating cancer antibody drug trastuzumab to polyethylene glycol (PEG). Applying force as pulsed ultrasound reduced the amount of PEG payload that the antibodies lost over a week from 50% to less than 10%.
Guillaume De Bo from the University of Manchester, UK, says that the study ‘nicely demonstrates the synthetic potential of polymer mechanochemistry’. ‘They have shown that mechanical force can be used to steer the reactivity of a molecule towards an otherwise kinetically disfavoured reaction pathway.’
References
W Huang et al,Nat. Chem., 2019, DOI: 10.1038/s41557-018-0209-2













No comments yet