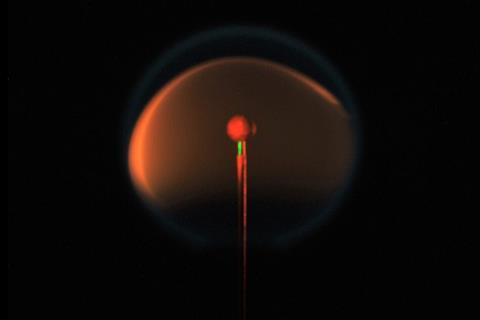
On board the International Space Station (ISS), astronaut Christina Koch is lighting candles to help scientists back on Earth resolve the long-standing question of why flames in microgravity produce less soot. Researchers hope that by understanding why soot formation is suppressed they can cut levels of it down here given that it is the second greatest contributor to global warming.
Diesel engines, stoves and coal-fired power plants produce an estimated 17 million tonnes of soot per year. Soot not only warms the planet two-thirds as much as carbon dioxide, it also pollutes cities – with detrimental effects on people’s health.
The nanoscopic particles are created during incomplete combustion, though how exactly this happens is still a matter of contention. Some scientists believe it’s the flow of gases in and around the flame that influences soot formation. Others, like Richard Axelbaum from Washington University in St Louis, US, think it’s all about the ingredients that go into a flame.

Axelbaum’s team has previously shown they can design cleaner flames. A regular flame burning in air – around 80% nitrogen and 20% oxygen – is sooty, but removing the nitrogen from the surrounding air and adding it directly into the flame’s fuel makes it burn soot-free. This is despite both flames burning at the same temperature.
The researchers are now working with ISS astronaut Koch to repeat the flame design experiments in microgravity. On Earth, candle flames emit a yellow glow from heated carbon particles. But in the absence of convection, like on board the ISS, candle flames become spherical and blue – meaning there’s less soot.
In a few weeks’ time, once the tests are completed, he hopes to find out whether his proposal is correct. If soot formation is dependent on the flame’s properties rather than gas flow, the designer flames should be as soot-free in microgravity as they are ones on Earth.
















No comments yet