A new, highly efficient electrochemical process converts propane into propene at room temperature. The team that developed the process believes that it is an economically viable, environmentally friendly approach to one of the most important reactions in chemistry.
Propene is widely used in the manufacture of numerous chemicals such as polypropylene, acrylonitrile and polyurethanes. The current thermal dehydrogenation process used to produce propene involves temperatures of up to 600°C and creates a mixture of gases that require subsequent separation. A more energy-efficient electrochemical method would therefore be extremely valuable, but previous efforts have achieved unsatisfactory activity or selectivity for propene.
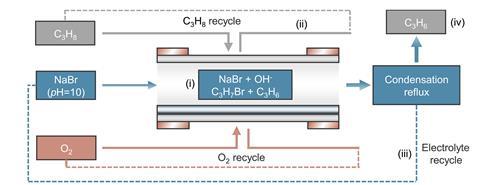
In the new work, researchers co-led by materials chemist Xiongwen David Lou at the City University of Hong Kong in China used an aqueous electrochemical cell containing sodium bromide. The anode was a gas diffusion electrode functionalised with a specially designed, self-assembling catalyst comprising hollow spheres of tin dioxide in an ionic liquid. The catalyst’s structure helps to concentrate bromine radicals – generated by the oxidation of bromide ions – and the propane feed gas, which react to produce bromopropane.
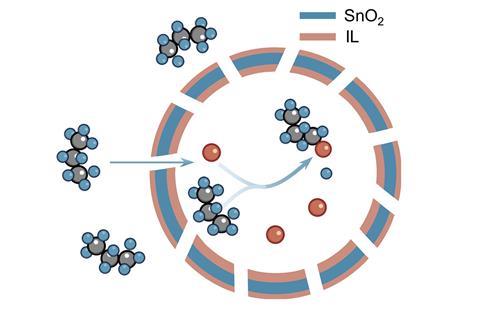
‘The alkyl chains of the ionic liquid create a hydrophobic interfacial environment that stabilises bromine radicals by reducing water molecule density near the catalyst surface, thereby suppressing unwanted radical quenching reactions,’ explain Lou and his colleague Jiarui Yang in emailed comments to Chemistry World.
Hydroxide ions generated at the cathode then react with the bromopropane, hydrolysing the carbon–bromine bonds to release propene and water, and regenerating the bromide ions.
The researchers found that the selectivity of the cell for propene was over 98%. This selectivity did not degrade over 6000 hours on stream, unlike in thermocatalytic reactions that show progressive coking of the catalyst. The researchers have conducted a techno-economic analysis and believe it could be commercially competitive with the current process. ‘The most pressing issue is scaling up, moving from our lab-scale electrodes to pilot-scale manufacturing,’ they note.
US-based chemical engineers Marcel Schreier at University of Wisconsin–Madison and Brian Tackett at Purdue University in Indiana are both impressed by the results.
‘It is a very nice demonstration, the rates are extremely high, it’s very impressive in that regard – and then there is this very interesting behaviour of this ionic liquid that they have on the anode,’ says Schrier, who adds that in his view ‘some of the mechanistic insights are a little bit preliminary’.
The most significant aspect in Tackett’s view is that the researchers separate the gaseous propane input from the gaseous propene output, although he notes that it’s not entirely clear to him how the experimental setup enables this. ‘If that’s accurate, this is a very nice demonstration, with reasonable rates of reaction, of electrochemically driven dehydrogenation of propane to propene, essentially without the need for extensive separations.’
References
J Yang et al, Science, 2026, DOI: 10.1126/science.aed2309















No comments yet