Deuterium can be electrochemically separated from ordinary hydrogen far more efficiently than current techniques using a ‘through barrier’ effect. The work, which achieves a record-breaking separation factor at room temperature, could be useful to the nuclear industry.
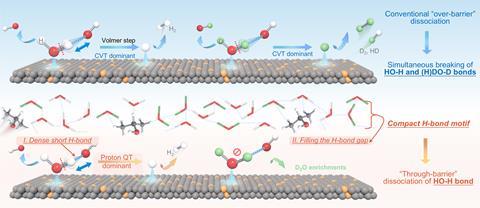
Deuterated water has many uses, ranging from a coolant in nuclear reactors to a drug for Huntington’s disease. However, it naturally makes up only about 120 parts per million of water and separating it from water containing light hydrogen (protium) is difficult. Owing to the mass difference, researchers have developed distillation and selective adsorption technologies, but these are both energy intensive.
A lower-energy alternative is electrochemical separation of an aqueous solution to discharge hydrogen at the cathode. ‘The lighter isotopes have a lower energy barrier for different reactions,’ explains electrochemist Magda Barecka at Northeastern University in Boston, Massachusetts. ‘In an electrochemical cell there would be multiple ways in which light water is reacted and therefore heavy water is enriched.’ Unfortunately, any chemical process that reduces the activation energy for cleavage of the bond between oxygen and protium also reduces the activation energy of oxygen–deuterium cleavage.
In the new work, the researchers took an alternative tack, focusing not on lowering the energy of bonds but raising them. Simulations suggested that adding isopropanol to an alkaline electrolyte reconfigured the hydrogen-bond network around the active sites in a ruthenium–nickel carbide electrode, making it more ordered and shortening the water’s bond lengths. When a potential difference was applied to the solution, the traditional chemical mechanism involving electron transfer to a loosely-bound proton was suppressed. However, protium – being much lighter than deuterium – has a more delocalised wavefunction. It therefore has a much greater probability of tunnelling through an energy barrier that it is classically forbidden from penetrating, and shortening the bond length makes this more likely.
The researchers found that their techniques’ ‘H2O separation factor’ (the ability to select for one type of water over the other) was more than twice as high as that of the best chemical sieving membranes. They designed a five-stage reactor and found that, at room temperature and a potential of just 0.4V, they could start with water and continuously enrich it to a deuterium atomic fraction of 80%. The process was even more selective at low temperatures, but its effectiveness at room temperature made this cooling uneconomic. ‘Subsequent coupling with nuclear power plants to enrich deuterium and tritium from their effluent using electrochemical hydrogen isotope separation technology could lead to a significant reduction in costs,’ the researchers from Hunan and South Central universities in China write.
‘The extent of the isotope separation achieved here is, to the best of my knowledge, significantly higher than the state of the art,’ says Barecka. ‘The authors are definitely proposing something very interesting and potentially scalable in terms of separating deuterium. It would be very interesting to see other labs repeating this research, using different ways to quantify isotope separation and going more into analytical chemistry, and then moving towards maybe a pilot-scale demonstration… Really showing that this is something that can be continuously achieved at scale.’
References
G Wen et al, Proc. Natl. Acad. Sci. USA, 2026, 123, e2533803123 (DOI: 10.1073/pnas.2533803123)













No comments yet