Researchers in Japan have combined two of chemistry’s most well-known structural motifs to make a new class of interlocked supramolecular system: ‘MOFaxanes’.

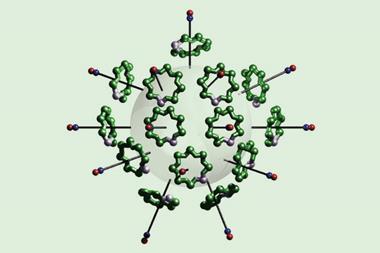
The new structure is a combination of a metal–organic framework (MOF) and a rotaxane. MOFs are crystalline materials formed of a network of metal ions and organic linker molecules. They generally contain nanometre-sized pores, which can be used to coordinate small guest molecules. These properties give MOFs a wide range of potential applications, such as in catalysis, gas storage and separation processes.
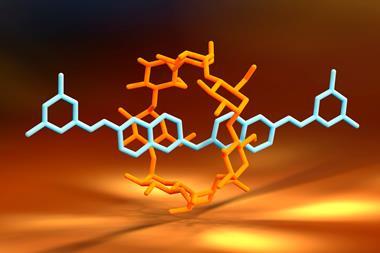
Meanwhile, rotaxanes are a class of interlocked molecules, in which a dumbbell shaped molecule is threaded through a macrocyclic ring. They are among the most famous examples of molecular machines, which were the focus of the 2016 Nobel prize in chemistry.
Now, a team led by Nobuhiko Hosono and Takashi Uemura from the University of Tokyo has combined the two ideas by threading polymer chains through the pores of a copper-based MOF. The researchers explained that they want to reverse the ‘conventional size relationship between a MOF and its guest’ by using ultralong polymers as the guest molecules. They note that MOFs’ tuneability means that in future it should be possible to make a much wider range of MOFaxane structures.
References
T Iizuka et al, Nat. Comms., 2023, DOI: 10.1038/s41467-023-38835-5














No comments yet