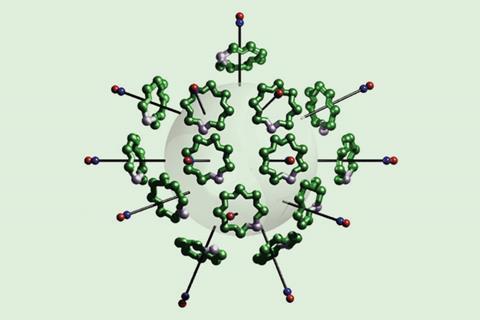
A formal 1014 rotaxane has been synthesised by decorating a polymer bead with paramagnetic complexes using click chemistry.
Rotaxanes are an unusual type of molecular architecture in which a macrocycle is threaded and trapped on a dumbbell-shaped molecule to create an interlocked complex. The macrocycle unit can freely rotate around the central axle, enabling the rotaxane to act as a type of molecular machine.
Magnetic particles have wide-ranging applications in structure determination techniques such as NMR but usually require a solid support such as functionalised polymer beads. Attaching many paramagnetic groups to a single bead could lead to additional uses, for example as contrast agents or ferrofluids, which need stronger magnetic properties.
An international team have now used click chemistry to functionalise 115µm polystyrene beads with many hybrid inorganic–organic [2]rotaxanes. {Cr7Ni}-pivalate rings were threaded onto an ammonium cation template molecule and capped with Tempo, an organic compound containing a free radical. A copper-catalysed click reaction then united these rotaxanes with the azide-functionalised polystyrene beads.
As each rotaxane unit includes a free radical, the team could also determine how many had bound to each polymer bead. Using electron paramagnetic resonance spectroscopy to detect unpaired electrons, they calculated that the surface of each bead accommodated 1014 rotaxane molecules, making these beads the largest formal rotaxanes reported.
In future, the team hopes to investigate the synthesis and applications of similar highly paramagnetic microparticles.
References
D Asthana et al, Commun. Chem., 2022, 5, DOI: 10.1038/s42004-022-00689-1













No comments yet