Visualising atoms’ wave function lets chemists tell true from indirect hydrogen atom transfer
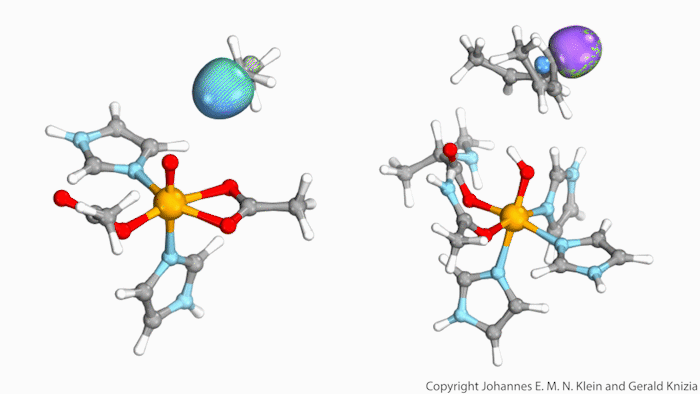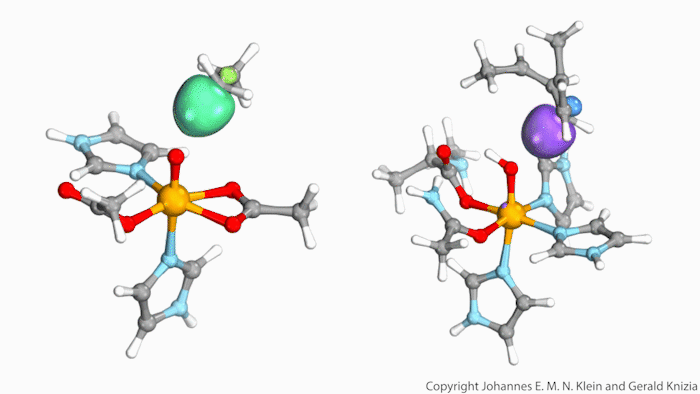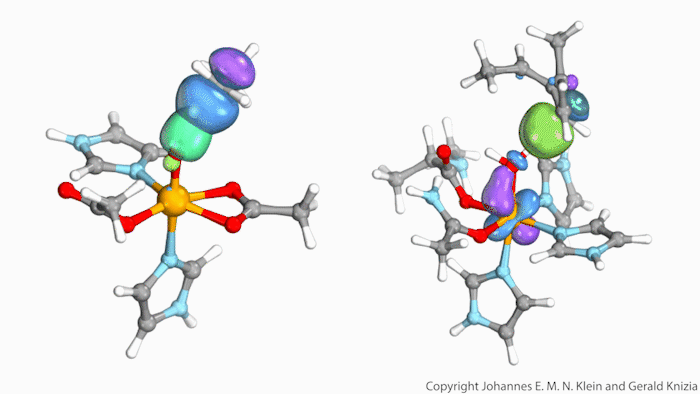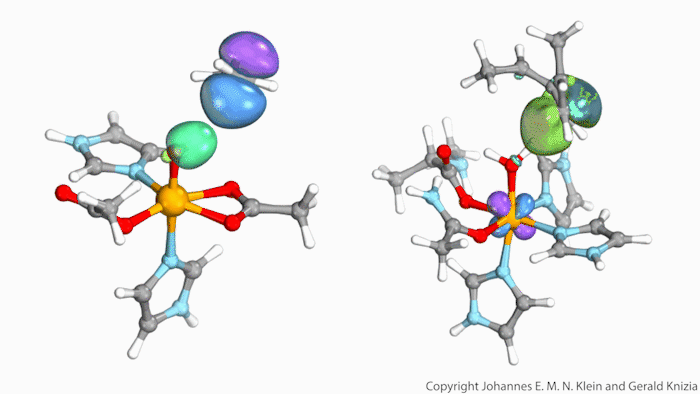
Watching computed orbitals – visual representations of atoms’ quantum dynamics – dance around during a reaction might help chemists distinguish between different types of hydrogen atom transfers. This might save them from having to run kinetic experiments and build complicated models based on the results.
In many biochemical reactions, as well as during hydrocarbon combustion, carbon–hydrogen bonds are broken through proton coupled electron transfer. In this net hydrogen atom transfer, the proton and the electron can either travel together or separately.
Finding out which mechanism a reaction prefers is essential if scientists want to modify or reproduce biological cascades such as photosystem II, which is responsible for water splitting during photosynthesis. However, uncovering how a reaction proceeds is not straightforward, and often requires gathering extensive experimental and computational data.
Johannes Klein from the University of Groningen, Netherlands, and Gerald Knizia from Pennsylvania State University, US, think that their intrinsic bond orbitals can help. These are calculated from the molecules’ wave functions and show where electrons are located. Snapshots taken at different times during the reaction allow chemists to distinguish between hydrogen transfer mechanisms by simply observing the orbitals’ shape and position.
The team tried out this method on two simplified biomolecules: lipoxygenase and taurine dioxygenase, two iron-containing enzymes that cleave carbon–hydrogen bonds. Lipoxygenase’s electron is seen travelling independently of its proton, though both move at the same time. In the dioxygenase the entire hydrogen atom moves as one.
References
J E M N Klein and G Knizia, Angew. Chem., Int. Ed., 2018, DOI: 10.1002/anie.201805511
















No comments yet