Alpha nucleophiles’ ability to get close to electrophiles – rather than their intramolecular lone pair repulsion – gives them unusual reactivity
The origin of the alpha effect, which gives some nucleophiles unusually high reactivity in substitution reactions, is one of chemistry’s unsolved mysteries. Now, computational chemists have uncovered a largely overlooked mechanism that provides answers to the problem. ‘Our breakthrough was formulating a causal relationship between the electronic structure of the alpha nucleophiles and their intrinsic reactivity,’ says lead researcher Trevor Hamlin from the Vrije University in Amsterdam, the Netherlands.
The alpha effect was first described in 1962 as the enhanced nucleophilicity of an atom – often oxygen, nitrogen or sulfur – due to the presence of an adjacent (alpha) atom featuring a lone electron pair. In the hydroperoxide (OOH–) and hydroxylamine (H2NO–) anions, for example, ‘reactivity is significantly increased without affecting overall basicity’, explains Joaquín Barroso-Flores, a computational chemist at the National Autonomous University of Mexico who wasn’t involved in the study. But ‘the electronic origin of the alpha effect has challenged chemists for many decades,’ he says.
Hamlin argues that previous explanations failed to explain the alpha effect because they only focused on the repulsion between the lone electron pairs on the nucleophilic and the alpha atom. ‘This mechanism should also increase the basicity and, therefore, fails to fully explain the alpha effect,’ he says.
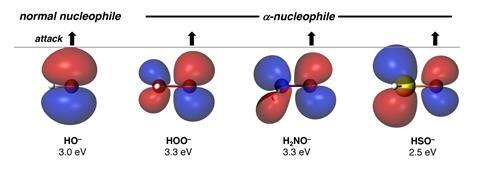
His team’s calculations use quantum mechanics to unveil a missing link. ‘Nucleophiles that exhibit the alpha effect engage in less destabilising steric repulsions with the substrate,’ says Hamlin.
Usually, interactions between two occupied orbitals have a destabilising effect. But the researchers’ simulations suggest that alpha nucleophiles feature significantly smaller orbital lobes in their Homo (highest occupied molecular orbital). ‘The alpha heteroatom polarises the Homo at the nucleophilic centre, reducing its size and lowering the steric repulsion,’ explains Barroso-Flores. ‘It also increases the energy of the Homo of the nucleophile, … promoting a more efficient overlap, which ultimately leads to an increased reactivity.’
‘This work provides a thought-provoking conceptual explanation on the origins of the alpha effect,’ says Igor Alabugin, an expert in structure and reactivity at Florida State University, US. However, ‘the theoretical analysis is simplified by… looking at only the anionic alpha nucleophiles,’ he comments. ‘One can expect that the analysis of neutral systems, where the alpha effect is only activated at certain geometries, would be more complicated.’
Barroso-Flores is also prudent. ‘[The authors] have found a very clear correlation between the structure of the nucleophile and the intensity of the alpha effect, however we must be cautious when invoking a causal relationship,’ he says.
Nevertheless, both experts value the contributions of this work to the understanding of the alpha effect. ‘[It] presents a thorough systematic analysis of alpha effect in a broad range of anionic systems at the same level of theory, and clearly shows [this] effect is not a general phenomenon,’ explains Alabugin. Moreover, he says, the study establishes a classification for different nucleophiles: some exhibit the alpha effect, others don’t and a third type exhibits an inverse alpha effect – a phenomenon in which adjacent heteroatoms decrease nucleophilicity.
‘The physical principles provided allow to qualitatively predict if a nucleophile possesses the alpha effect solely based on its intrinsic electronic structure,’ says Hamlin. ‘Therefore, [this] has the potential to enable chemists to better predict the reactivity of fundamental reactions.’
References
T Hansen et al, Angew. Chem. Int. Ed., 2021, DOI: 10.1002/anie.202106053

















No comments yet