
A counterintuitive technique developed by researchers in the US and South Korea allows pentametallic nanoparticles of relatively uniform size and composition to form spontaneously from a precursor solution containing the five metals. The nanoparticles are a promising catalyst for the decomposition of ammonia into hydrogen and the technique could also potentially be extended to produce other multimetallic nanoparticles.
Multimetallic nanocrystals can sometimes offer catalytic properties that nanocrystals of a single metal cannot such as higher atom economy for a precious metal or synergistic interactions between the metals. However, they can be difficult to synthesise because different reactivities or natural crystal structures of the constituent metals may not lead to compositionally uniform products. Most work to date has focused on energy-intensive schemes in which mixtures of the metals are cooled from very high temperatures in milliseconds or even microseconds to kinetically trap them, preventing the metals from forming separate phases.
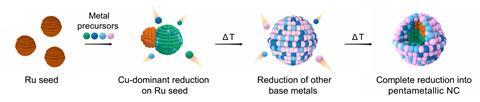
In the new work, researchers in the groups of Matteo Cargnello at Stanford University in California and Hee-Tae Jung at Korea Advanced Institute of Science and Technology took a subtler approach, depositing the metals from solution onto ruthenium nanoparticle seeds by mixing them with metal acetylacetonate precursor solutions and heating the mixture. When they studied bimetallic compositions, they found differing results. Iron formed variably sized, self-nucleated nanoparticles separate from ruthenium, copper formed core–shell nanoparticles, and cobalt and nickel both formed mixtures of the two.
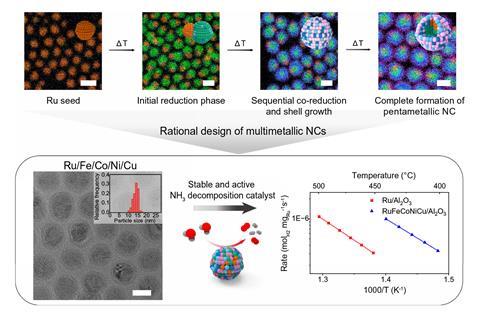
When the researchers added more metals together, however, they found that – instead of producing the expected morass of products – the distribution became more uniform. When they added precursors of all five metals, they found that they could reliably obtain RuFeCoNiCu particles about 20–25nm in size with constant atomic composition. Time lapse investigations indicated that the copper deposited first onto the ruthenium before other metals deposited onto the bimetellic nanoparticle. ‘We think ruthenium has more affinity and miscibility with the cobalt, and copper has more affinity and miscibility with the nickel,’ explains Cargnello. ‘The fact that you have already deposited both copper and ruthenium on the same nanocrystal gives you the right conditions to deposit both nickel and cobalt on the same nanocrystal, rather than forming more mixtures in solution, and it’s the same for the iron.’
At a temperature of 900°C, the multimetallic catalyst could achieve a catalytic rate four times as high as ruthenium alone for the decomposition of ammonia. Its effectiveness was not optimal under conditions that favoured ammonia synthesis, but the research was financially supported by BASF’s California Research Alliance Program looking at its potential in a hydrogen economy.
‘It’s an interesting work,’ says Peidong Yang at the University of California, Berkeley, who is the director of BASF’s California Research Alliance that approved funding for the project but was not involved in the research. ‘I think Matteo’s group managed to figure out this sweet temperature window that’s doing this sort of compositional focusing. It’s a little bit surprising to see this focusing effect because iron, nickel, cobalt, ruthenium – their precursor reduction chemistry is very, very different – and second, they’re not even in the same crystal structure.’ He says he bets that, at sufficiently high temperatures, the metals will phase-separate, but that this may not matter in practice. The ultimate question, he says, is whether or not the method proves to be generalisable to other systems. ‘If it’s a really universal phenomenon, I think this is quite significant,’ he says.
Update: Peidong Yang’s connection to the project was clarified on 13 May 2026
References
J Yoon et al, Science, 2026, DOI: 10.1126/science.aea8044
















No comments yet