A material with a self-destruction switch has been made by chemists in Germany. Similar to biological systems, the material self-assembles from a precursor as it consumes a chemical fuel and disappears after a pre-programmed time – properties that could help it carry drugs around the body and release them when and where they are needed.
Scientists have developed numerous materials that respond to stimuli like light by changing their shape or moving. However, these materials often only have an off-to-on response and some require a second input to switch back off. ‘For most applications that’s completely fine, but in biomedical applications it would be useful to have a material that switches itself back off after having been activated,’ says Job Boekhoven from the Technical University Munich, who led the new study.
Cells can move and divide by consuming chemical fuels like adenosine triphosphate (ATP). Unlike most artificial materials they are driven by kinetic rather than by thermodynamic parameters. ‘The crucial difference between most artificial supramolecular materials like liquid crystals and biological materials is that the former are essentially in equilibrium. This means they are in a very stable state and last for a long time, but they miss the dynamics of biological materials like cells,’ Boekhoven says.
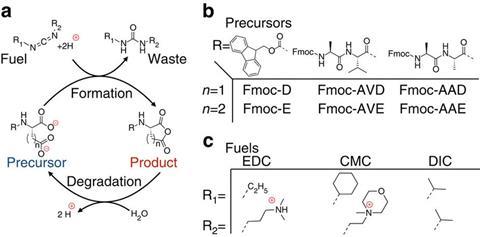
Inspired by this concept, his team designed a chemical system in which precursors, amino acid dicarboxylates, convert into anhydrides by using a carbodiimide fuel. Driven by hydrophobic interactions, the anhydrides assemble into large networks – and then self-destruct, reverting back to the precursor after minutes or hours depending on the amount of fuel. ‘Our materials are not in equilibrium; they don’t want to stay in that state but autonomously degrade back to the precursor state without the need for a second input,’ explains Boekhoven. The system can even be reused – adding another batch of fuel converts the precursor back into the anydride.
Depending on which and how many amino acid groups the precursor carries, the anhydrides assemble into different shapes: spherulites, colloids or fibres, though it is hard to predict which molecules will end up forming which networks. The researchers used spherulites to create self-erasing ink, while fibre-like material can be used for hydrogels with tuneable lifetime.
Boekhoven believes that non-equilibrium materials could make drug delivery easier: ‘The material could sequentially release several agents at specific times and with specific rates.’ Beatriu Escuder Gil, who works on bioinspired supramolecular materials at the Jaume I University, Spain, agrees. ‘I think applications in drug delivery are the most feasible.’ However, she adds, ‘the challenge is to go into more complex systems, coupling these with other reactions and make something more lifelike’.
References
M Tena-Solsona et al, Nat. Commun., 2017, DOI: 10.1038/ncomms15895




















No comments yet