For the first time the gap between manmade electricity and the biochemical energy that powers life, adenosine triphosphate (ATP), has been directly bridged using a new to nature metabolic pathway. This discovery discards the need for complex membranes and the roughly 3000 reactions used by cells to generate ATP in nature and consists of just four enzymes.
‘Manmade electricity didn’t exist for much of Earth’s history, and this is why nature probably has never learned to live off the electrical grid,’ said Tobias Erb at the Max Planck Institute for Terrestrial Microbiology in Marburg, Germany whose group built the synthetic pathway. The newly minted acid/aldehyde ATP (AAA) cycle improves on the current methods that use electricity to power bioengineered systems by eliminating the need for an intermediate molecule like hydrogen or the requirement to grow complex microorganisms directly on electrodes. Erb conceived of a simpler pathway, requiring a minimum set of reactions to capture electrons and convert this energy into ATP. Such a reaction could then serve as an adapter to convert electricity directly into ATP for a variety of purposes.
The first hurdle was finding an enzyme that accepts electrons directly and performs a simple redox reaction. Thankfully, the perfect candidate was serendipitously available from colleagues at the University of Marburg who were conducting basic research on a poorly understood bacterium, Aromatoleum aromaticum. Originally discovered 30 years ago, A. aromaticum produces a class of enzyme called an aldehyde oxidoreductase (AOR) which reduces an acid to an aldehyde, storing the electrical energy in this bond. With this achieved the AAA cycle only needs three further steps. ‘The remaining three enzymes are responsible for the regeneration of the aldehyde,’ explained Shanshan Luo, first author of the study published in Joule, ‘this process releases energy that is used to generate ATP’.
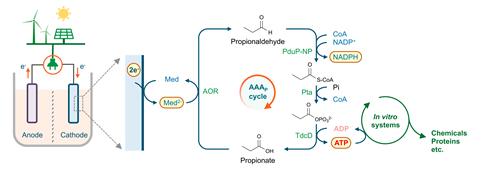
‘It is a fantastic piece of bioengineering research, and it closes a critical technology gap for engineering self-sustaining biological systems,’ said Steffen Lindner, a researcher at the Max Planck Institute of Molecular Plant Physiology in Potsdam, Germany. Potential applications for this technology include artificial photosynthesis and the storage of surplus energy from renewable sources. For Erb, however, driving protein synthesis directly from electricity was an exciting application they were able to achieve in the paper. Jixun Zhan, a professor in the department of biological engineering at Utah State University agrees. ‘They are many biosynthetic reactions that require ATP consumption,’ says Zhan. ‘One application of the AAA cycle is to power natural product biosynthesis.’
The plug and play nature of the AAA cycle means it will have a wide range of potential applications, however there is lots of research still required. According to Lindner, ‘transferring [the AAA cycle] into an industrial or commercial application will require the optimisation of the process with regards to cost, stability, longevity, and the identification of economically viable products and applications’. Erb agrees and says that now other researchers can take this work and begin to develop the specific hardware components needed to harness the cycle for applications.
References
S Luo et al, Joule, 2023, DOI: 10.1016/j.joule.2023.07.012










No comments yet