The first direct evidence of nitrogen fixation from volcanic lightning has been found by a team of researchers from France and elsewhere. This work, which reinforces previous laboratory studies and theoretical models, supports models of prebiotic chemistry in which volcanic lightning plays a crucial role in synthesising nitrogen for essential prebiotic reactions.
The dinitrogen triple bond is one of the most stable in chemistry. Although the atmosphere comprises around 78% nitrogen, only a few nitrogen-fixing bacterial enzymes can break it and convert it into a bioavailable form. Other organisms obtain their nitrogen from the work of these bacteria. Nitrogenous fertilisers – mainly derived from ammonia – are also added artificially by humans. For life to emerge, however, bioavailable nitrogen must have emerged without life, and one of the principal suspects has long been lightning strikes.

Explosive eruptions cause spectacular lightning displays owing to charge separation in the atmosphere by the movement of rock, ash and ice particles. The 2022 Hunga Tonga underwater eruption near Indonesia produced around 400,000 lightning strikes in six hours. The intense energy in the arc discharge can break chemical bonds but the exact mechanisms by which atmospheric nitrogen could be fixed by an eruption remain unclear. ‘When volcanoes degas, they do not release much nitrogen,’ says Erwan Martin at Sorbonne University in Paris. ‘Sometimes they have N2 but it’s in very tiny amounts.’
Volcanoes do, however, release huge amounts of sulfur and halogens. Martin and colleagues therefore studied identifiable volcanic deposits from geologically recent eruptions when the Earth had an oxidising atmosphere. They found that the samples were unexpectedly rich in nitrate. The concentration correlated well with the concentrations of sulfate and chloride ions, suggesting that they had a common origin. Moreover, the quantity of nitrate was just as high in the non-porous rock produced by the solidified pyroclastic flow as the pumice produced from fallout deposits. The researchers concluded, therefore, that the nitrate had not been introduced by atmospheric processes over time but had been present when the rock was formed.
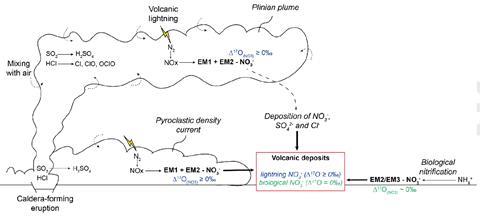
This did not explain how nitrate was produced, however. The researchers believe that the exact mechanism may vary, but that the general mechanisms all involve the breaking of the dinitrogen bond in the atmosphere to create nitrous oxides, which are then oxidised to nitrate and fall to Earth. Under the more reducing atmosphere prior to the great oxidation event after photosynthetic plants emerged, this would not have occurred. ‘The main process is to break down the N2 double bond,’ says Martin. ‘Then you can add to it oxygen, you can add to it hydrogen… The main thing is that this molecule could be assimilated by life.’
Cosmogeochemist Jeffrey Bada of the University of California, San Diego, who has previously argued that volcanic lightning on the early Earth could have proved crucial to the synthesis of amino acid precursors, says: ‘I think the idea that volcanic lightning is a process by which you can convert N2 into usable compounds for biology is an important one and with their new study they have sort of confirmed that in a more rigorous way.’ He adds, however, that, ‘I think if they’d wanted to make a bigger splash they’d have tried to convert this into what the early Earth was like – this idea that this happened on the early Earth is kind of cited in passing at the end of the paper.’
Update: The headline was changed on 13 February 2024 to make explicit that volcanoes on early Earth could have generated a range of nitrogen compounds that could have contributed to prebiotic chemistry
References
A Aroskay et al, Proc. Natl. Acad. Sci. USA, 2024, 121, e2309131121 (DOI: 10.1073/pnas.2309131121)















No comments yet