Mixed valencies and overlapping π-electron clouds help electrons move from molecule to molecule
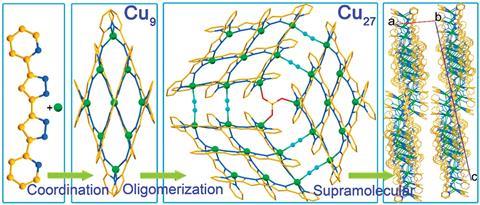
A new supramolecular architecture whose peculiar one-dimensional framework results in crystals with unusually high electrical conductivity has been made by researchers in China and France.
Jing-Lin Zuo of Nanjing University and his team first set out to investigate spin effects in polynuclear metal complexes – but after attempting unsuccessfully to create a grid-like organometallic cluster made from Fe(II), they decided to investigate other metal complexes.
Zuo says copper was an obvious alternative due to the large range of coordination geometries that copper can adopt. They combined nine organocopper complexes to construct a Cu9 coordination compound, then induced oligomerisation to combine three Cu9 units into a flat Cu27 cluster with a mixture of Cu(I) and Cu(II) atoms. Isolating the Cu27 cluster resulted in lustrous silvery crystals, and after connecting the crystals to a circuit, Zuo’s team were surprised to find that they were remarkably electrically conductive.
Zuo terms this a hierarchical tandem assembly, where three important sequences of events happen one after another, resulting in a big supramolecular architecture with interesting properties. ‘It’s like a three-wheeled bicycle, and it works if all three are in motion,’ Zuo tells Chemistry World. He says that the flat Cu27 clusters stack like dinner plates to form crystals, and they could potentially serve as one-dimensional conducting solids. ‘There are multiple advantages – we have a cluster of 27 copper atoms, where each one is a redox-active centre – allowing a potential manipulation with 27 electrons, compared to an organic molecule which only has two,’ Zuo continues.
When asked why the electrical conducting properties of the material are so good, Zuo emphasises that the ease by which electrons move from molecule to molecule is key. ‘This happens because of the mixed valency, which means that vacant positions in the orbitals of neighbouring molecules can accommodate moving electrons,’ he says. ‘But this alone isn’t sufficient. In quantum chemistry jargon, we need additionally overlapping π electrons from the surrounding organic ligands to create the pathways to allow a moderate transfer integral.’
He likens this to a sheet with wells, each filled with water. ‘If we shake the sheet so that the depths of the wells are altered, then the water (or electrons) will move over the deeper ones. This causes the water to move across them. In our case, the electrons from each cluster (or well) move from cluster to cluster, and the motion of electrons is equal to an electric current.’
Zuo says that the lightweight, layered structure could be easily deposited over a large surface, which could pave the way to high-performance electronic devices.
‘Zuo’s hierarchical assembly of functional units begins to move the field along the pathway to functional devices,’ comments Jonathan Steed, a supramolecular chemist at Durham University in the UK. However, he adds that it needs more work before it can be used in practice: ‘Supramolecular grid structures are well known, but their early promise in delivering novel properties, with long-term applications in electronic devices, has proven difficult to realise.’















No comments yet