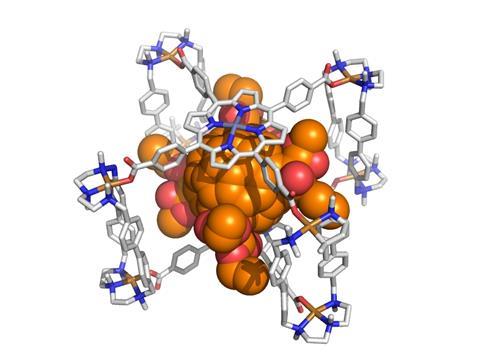
A supramolecular mask makes it easier to perform selective reactions on fullerene compounds. This could help chemists developing solar cell materials to investigate the properties of functionalised C60 molecules.
Compounds based on the famous buckminsterfullerene structure have the potential to improve the efficiency of solar cell materials or even be turned into drugs. As such, researchers are keen to make variants of these structures to try and fine-tune their properties. But doing selective chemistry on substrates with 60 identical sites is no easy feat, with most syntheses requiring laborious protecting-group strategies and impractical separations of unwanted byproducts.
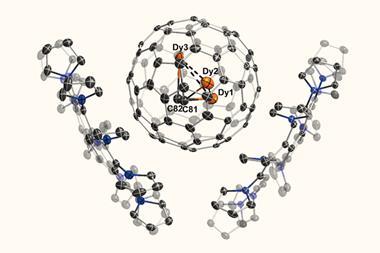
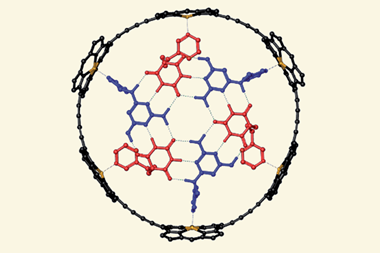
Now, researchers in Spain have created a supramolecular mask that allows them to perform selective chemistry on a range of fullerene substrates. The porphyrin-based structure straddles the fullerene, limiting the number of carbon atoms available for further reactions. This effectively acts like a stencil, allowing chemists to perform addition reactions at specific sites on the fullerene.
As the supramolecular mask has a higher affinity for the fullerene than the modified fullerene compounds, the products can be released from the mask by simply stirring them with an excess of starting material.
The researchers demonstrated their method by carrying out a range of Bingel–Hirsch cyclopropanation reactions. The researchers say their work provides an important new way of making ‘unprecedented pure-isomer poly-functionalised C60-based compounds’ for testing in solar cell devices.
References
C Fuertes-Espinosa et al, Chem, 2019, DOI: 10.1016/j.chempr.2019.10.010





![Terphen[3]arene](https://d2cbg94ubxgsnp.cloudfront.net/Pictures/380x253/7/3/6/518736_d2cc00040gf1_733670.jpg)








No comments yet