Researchers in Japan have synthesised nanoporous ultra-high-entropy alloys that contain 14 elements. Tests showed the alloys were capable of splitting water and performed better than commercial platinum-based hydrogen evolution reaction catalysts and iridium-based oxygen evolution reaction catalysts.
‘Most conventional alloy catalysts contain a primary metal constituent with high atomic percentage, such as platinum, and one or two kinds of minor metal constituents with relatively low atomic percentage,’ explains Guofeng Wang, a materials scientist at the University of Pittsburgh, US, who was not involved in the study. ‘The minor metal constituents normally provide beneficial ligand or strain effects to enhance the catalytic performance of the primary metal constituent.’ High-entropy alloys, however, are a relatively new concept and contain five or more different elements in near-equiatomic proportions.
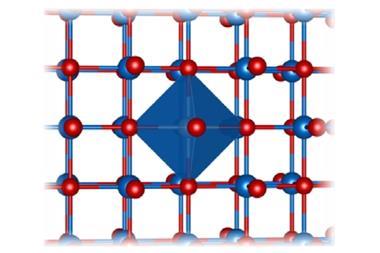
Reaching far beyond five elements, Takeshi Fujita, of Kochi University of Technology, and his team from across Japan have synthesised nanoporous ultra-high-entropy alloys containing 14 different elements, namely, aluminium, silver, gold, cobalt, coper, iron, iridium, molybdenum, nickel, palladium, platinum, rhodium, ruthenium and titanium, via a simple one-step dealloying process.
‘The reported ultra-high-entropy alloy catalyst has a nearly equimolar composition for all the constituents,’ notes Wang. ‘Therefore, there is no distinction between primary and minor constituents anymore.’
High-entropy alloys composed of so many different elements are unusual, but Fujita says it is the high degree of synergy within these alloys that leads to their distinct properties, such as their ‘high configurational entropy, lattice distortion and sluggish diffusion’, as well as their potential applicability as omnipotent catalysts.
‘The active surfaces of high-entropy alloys consist of millions of different possible atomic arrangements,’ says Fujita. ‘Therefore, some local atomic pairs may unexpectedly show excellent catalytic properties.’
Fujita notes that synthesising ultra-high-entropy alloys containing so many elements of equal distribution is not always straightforward. ‘Like water and oil, it’s not easy to mix many elements uniformly,’ he explains. ‘[However], we found a simple way to mix 14 elements uniformly [via] a simple technique, alloying with aluminium and dealloying aluminium by etching.’
The nanoporous structure introduced by the dealloying process increases the surface area and the number of potential active sites within the alloys, improving their catalytic performance. Moreover, the distinct multimetallic composition of the alloys can facilitate charge transfer and the adsorption or desorption of various ions.
Cocktail effect
Fujita describes the 14-element composition as having a cocktail effect because mixing so many different chemical components alters their behaviour. ‘The cocktail effect does not come from the sum of the elements, but [from] the multiplication of elements. We know that the cocktail is always tasty not because of the sum of each element, but additional unexpected tastes from the combination of elements.’
The team evaluated the electrocatalytic performance of their nanoporous ultra-high-entropy alloys in the hydrogen evolution reaction and oxygen evolution reaction under acidic conditions. They showed excellent electrochemical stability, rapid electron transport and high reaction kinetics. Characterisation confirmed that the alloys mostly maintained their morphology and porous structure, even after spending 10 hours in an acidic environment.
While the catalytic performance of the nanoporous ultra-high-entropy alloys is largely attributed to the synergistic effects of the 14 elements, it is far too complex to define due to the numerous possible element combinations within the alloys.
Wang notes that while the study will ‘motivate the wider community to examine the broader application of ultra-high-entropy alloys’, questions about this synergistic effect and the underlying mechanism regarding the electrochemical stability remain.
And while Fujita’s team selected 14 major elements commonly used as catalysts for their nanoporous ultra-high-entropy alloys, it may be possible to use even more elements in the future. ‘Non-metals, [such as] sulfur or phosphorus, could technically be used and they might lead to unexpected sulfides or phosphides for catalysis,’ remarks Fujita. However, ‘the maximum possible number of constituent elements in high-entropy alloys … remains an open question.’
References
This article is open access
Z-X Cai et al, Chem. Sci., 2021, 12, 11306 (DOI: 10.1039/d1sc01981c)










No comments yet