Photoredox catalysis enables site selective amination and could make production of anilines safer
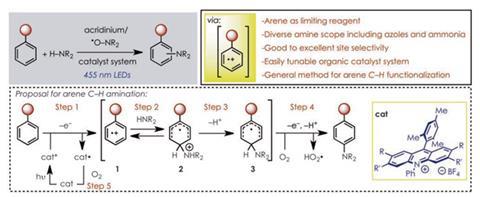
A new method for site selective amination of aromatic organic compounds has been developed by researchers in the US. The protocol, which does not require expensive catalysts, harsh conditions or pre-functionalised reactants, could be used to efficiently produce a variety of important agrichemicals, precursors and, above all, pharmaceuticals.
Aryl amine compounds are widely used as pharmaceuticals as the nitrogen lone pair provides a binding site for enzymes, allowing drugs to inhibit their activity. The techniques commonly used to attach a nitrogen atom to an aromatic ring require the C–H bond first be functionalised. In the Buchwald–Hartwig reaction, for example, an aryl halide and a primary or secondary amine are heated to around 100ºC with a palladium catalyst, causing cross coupling of the amine to the aromatic ring with the elimination of the hydrogen halide. Such pre-functionalisation steps lower the efficiency of the reaction, add to the expense and generate more waste. Furthermore, if the halide cannot be produced, then nor can the amine.
Synthetic chemist David Nicewicz and colleagues at the University of North Carolina have now developed a method to transform the C–H bond directly into a C–N bond using a modified photosensitive acridinium dye and 2,2,6,6-tetramethylpiperidine-1-oxyl as catalysts. On absorption of visible light, a powerful excited state oxidant is produced that removes an electron directly from an unfunctionalised benzene ring, forming a radical cation. The amine reacts directly with this radical cation and, after oxidation, the aryl amine is produced, with yields of up to 97%. ‘You can use the aromatic compound as you would pull it off the shelf. You can use the amine as you would get it off the shelf. You can use your catalyst mixture in the presence of oxygen and light and boom, you get your aromatic amine directly out of the reaction,’ says Nicewicz. ‘The only by-product is hydrogen peroxide.’
Among other compounds, the researchers could produce anilines using ammonia or an ammonium salt as the nitrogen source. Anilines, which are used in numerous industrial applications such as the production of urethane polymers, are currently produced industrially by nitration of aromatic compounds, followed by reduction. The nitrated aromatics can be highly explosive, with trinitrotoluene or TNT being a well-known example, so the production of anilines by this method is extremely hazardous. ‘This would get around the nitration reaction completely,’ says Nicewicz.
‘It’s very interesting for several reasons,’ says physical organic chemist Burkhard König of the University of Regensburg in Germany. ‘The number of direct conversions of an aromatic compound into an amino-functionalised compound by activating a C–H group is, in my opinion, still limited and this is a fantastic expansion of the methodology.’










No comments yet