Humanity took a long time to notice the second most common chemical element in the universe, James Mitchell Crow notes. Today we would struggle to do without it
The voyage that French astronomer Pierre Janssen undertook from Europe to the eastern Indian province of Madras during the spring of 1868 was no overnight endeavour. Janssen was travelling half-way around the world in the hope of becoming the first person to observe the upcoming solar eclipse using a hot new scientific instrument called the spectroscope. Using this new instrument, he hoped to make new discoveries about the composition of the Sun – assuming heavy cloud cover didn’t spoil his view.
During his voyage, and the worrisome run of rainy weather that preceded the day of the eclipse, Janssen would have had plenty of time to ponder the scientific giants upon whose shoulders he hoped to stand. Ably anchoring this human pyramid was Isaac Newton himself.
The discovery that set Janssen’s mission in motion was Newton’s famous 1666 observation that a beam of sunlight, passed through a glass prism, spreads into a rainbow of colour. White light, Newton showed, was a mixture of all the other colours combined. As techniques for dispersing sunlight gradually improved, however, the solar spectrum turned out not to be the perfect array of colour it had first appeared. In 1814, German lens maker-physicist Josef von Fraunhofer directed the sunlight through a narrow slit before passing it through a glass prism – and showed Newton’s rainbow was riddled with holes.
It was a first glimpse of a new element
Fraunhofer’s thin slit set-up dispersed the sunlight more effectively than a prism alone could achieve, thereby revealing no less than 574 sharp, dark lines in the solar spectrum. But not until 1859 did fellow Germans Robert Bunsen and Gustav Kirchhoff figure out where the missing wavelengths had gone (see Chemistry World, September 2013, p35). Kirchhoff invented a prism-based instrument – the spectroscope – that the duo used to show that some of the dark lines in sunlight were an exact match for the characteristic pattern of lines in the light emitted when the salts of recently discovered metals such as sodium and potassium were burned in a flame.
The scientists showed that each element absorbed and emitted its own pattern of wavelengths, as unique as a fingerprint – an effect we now know is caused by electrons absorbing or releasing light to hop excitedly between energy states. Bunsen and Kirchhoff had proven that the Sun was composed of the same chemical elements found on Earth. Using Kirchhof’s spectroscope, they went on to discover two unknown elements, rubidium and caesium, by burning residues from mineral water samples.
The race was on to find more new elements using the spectroscope – which led, in 1868, to Janssen’s mission to point one at the Sun. By performing his experiment during an eclipse, Janssen hoped to see the elements present around the Sun’s fringe, in its atmosphere.
On the day of the eclipse, the clouds finally dispersed. As the eclipse commenced and darkness descended over Madras that 18th August, Janssen saw the by-then well-known emission lines of hot hydrogen – and a bright yellow line never previously seen. It was a first glimpse of a new element, Janssen concluded.
As it soon turned out, you didn’t need a solar eclipse to see this striking yellow line. An overcast autumn day in the UK university city of Cambridge would also do. On the 20th October that year, unaware of Janssen’s observations, English astronomer Norman Lockyer saw the yellow line too – and decided to name this unknown chemical element helium, after the Greek word for the sun, helios.
The two men’s papers on their observations arrived at the French Academy of Sciences in Paris on the same day, and so they shared the credit for discovering a new element.
Which was when several decades of trouble for the pair began.
Extending the periodic table
Not everybody was convinced Janssen and Lockyer had discovered a new chemical element in the fringes of the Sun. Some scientists greeted the idea of the celestial element with outright ridicule, which only grew as the years passed and no trace of helium was found on Earth. Even Lockyer’s own assistant thought the yellow line was a probably previously undetected emission of hydrogen.1
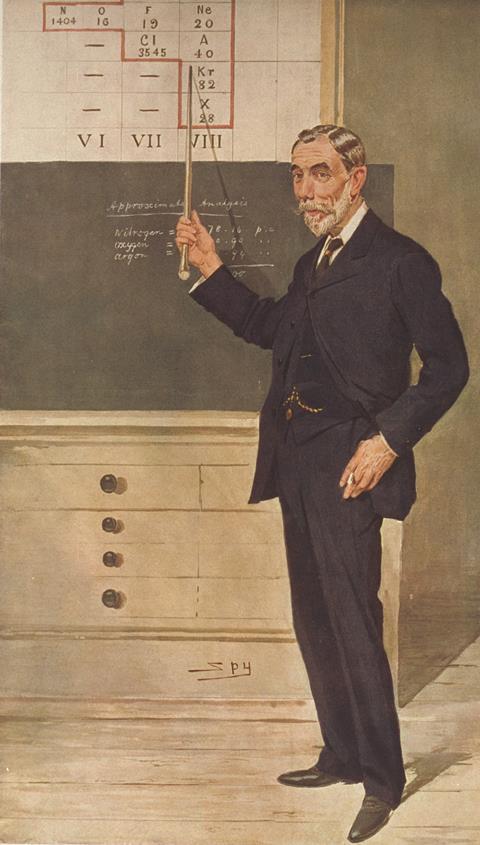
Prominent among non-believers was a rather formidable opponent, Dmitrii Mendeleev. The Russian chemist was no stranger to hostility and derision, having been on the receiving end for more than half a decade following his 1869 publication of his periodic table. But for Mendeleev, spectacular vindication of his discovery came in 1875, when – using a spectroscope – Paul-Emile Lecoq de Boisbaudran discovered gallium, an element whose atomic weight fitted perfectly in a gap in the table. The discoveries of scandium and germanium soon afterward plugged further holes. But none of these aided the case for helium, because the periodic table of the time had no gap for helium to fill.
It was an omission that would come back to haunt Mendeleev. In 1894, English chemist William Ramsay discovered an unreactive gas, which he named argon, released by heating a sample of a mineral. Unsure what to make of this new element that his periodic table had not predicted, Mendeleev argued Ramsay had not discovered a new element at all, but a previously unobserved three-atom molecule of nitrogen akin to ozone (O3).
But in 1895, repeating the heating experiment with a uranium mineral, Ramsay isolated another inert gas. This one produced a spectral line that was an exact match for those Janssen and Lockyer had observed in the Sun. Ramsay had found helium on Earth. Ramsay soon went on to discover krypton, neon and xenon – all remarkably unreactive. The completed new set of elements convinced Mendeleev that the periodic table needed expanding.
The noble gases, headed by helium, now sit on the far right hand side of the table, a position that reflects their neatly filled outer shell of electrons. This very stable electron arrangement is the reason for these element’s remarkably low chemical reactivity, with helium the most unreactive element of them all.
Ramsay’s discovery of a whole new family of elements that needed bolting on to the right side of the periodic table won him the 1904 chemistry Nobel prize. That same discovery, some have argued, simultaneously scuppered Mendeleev’s own chances of winning the award.1 Despite his remarkable contribution to science, the Russian chemist never received the Nobel.
Up up and away
It was no lucky accident that Ramsay isolated helium from a uranium-rich mineral. It is the slow decay of radioactive uranium and thorium nuclei that is the main source of helium on the planet today. Several uranium and thorium isotopes decay by emitting alpha particles – a little cluster of subatomic particles consisting of two protons and two neutrons. This combination of subatomic particles is the very set that makes up the helium nucleus.
Across the universe, helium is the second most common element after hydrogen. Most of this helium originates from a brief period a couple of minutes after the Big Bang, when temperatures cooled enough for small clusters of subatomic particles to form the first atomic nuclei. Some 380,000 years later, the universe finally cooled enough for these nuclei to capture electrons, forming the first atoms – mainly hydrogen and helium, which still dominate the universe today.
On Earth, however, it’s a different story. Helium is so light it is not held down by Earth’s gravity. Being inert, it is not held down by chemistry either. The helium present when the Earth was forming has drifted away into space. Jupiter and Saturn, bigger planets with stronger gravity, held on to their helium and it is a major component of the gas giants’ composition. As Lockyer and Janssen observed, it is also a major component of the atmosphere of the Sun.
When Ramsay discovered the noble gases, some members of the family very quickly found uses. One of the first experiments the chemist performed on newly isolated neon at his University College London labs was to excite it with an electrode. The gas gave off a stunning red glow that was soon lighting up the streets of Paris and Los Angeles in the form of advertising signs.

Helium took a little longer to find a use. The process was eventually hastened by the outbreak of the first world war.2 Germany’s military zeppelin balloons focussed UK and US military attention on lighter than air gases. Helium couldn’t quite match the lifting power of hydrogen, but it had the distinct advantage that it was chemically inert and would not burn if struck by enemy fire. A decade or so before the outbreak of war, helium had been discovered to be naturally present in some deposits of natural gas, notably in Texas. When the US entered the war, the Bureau of Mines issued contracts for the construction of three helium extraction plants.
With airships soon superseded by aeroplanes for military and commercial flight, helium’s use as a lighter than air gas did not quite take off as expected – although today it is used to fill balloons for high altitude scientific research, as well as for party balloons. For decades the US produced far more helium that it used, which it corralled underground in depleted gas fields.
In 1996, to recover the sizable debt it had accumulated buying up and storing all this helium, the US government announced a grand sell off of the stored gas – to the great concern of scientists. By this point, demand for helium was rocketing. Its inert nature had seen it adopted for creating a protective atmosphere when making fibre optics and semiconductors, and for arc welding, for example. Much more significantly, helium had become indispensible for modern medical imaging machinery, and well as to helping us probe the deep mysteries of the cosmos. Because helium has the lowest boiling point of any element, an ultra-chilly –269°C, it is an essential coolant for the very low temperatures needed by the superconducting electromagnets used in hospital MRI machines. For the same reason, it cools the magnets of the Large Hadron Collider, as well as some NMR machines.
Once helium escapes into the air, this light gas is gone forever. By 2015, the British Medical Association was concerned helium supplies would soon need to be regulated to guarantee supply of the gas into the medium and long term. But that year, a team from the UK universities of Oxford and Durham, working with helium exploration company Helium One, used a new method of systematic search to discover a natural helium resource for the first time.

The huge helium find is in the Rift Valley in Tanzania, where helium seeps had first been identified in the 1950s. The team used techniques for oil exploration, assessing factors such as rock type – but showed that the key to adapting the techniques for helium exploration was to factor in the presence of nearby active volcanoes. ‘Volcanic activity likely provides the heat necessary to release the helium accumulated in ancient crustal rocks,’ said Diveena Danabalan, a member of the Durham team. But there’s a goldilocks dimension to the volcano’s location, she adds. ‘If the gas traps are too close to a given volcano, they run the risk of helium being heavily diluted by volcanic gases such as carbon dioxide’.
Some of the trapped gas the team has sampled is between 8% and 10% helium, a far higher proportion than the helium sites currently exploited.3 According to Helium One, current estimates put the amount of helium present in the Rift Valley sites surveyed so far at almost 100 billion cubic feet (2.8 billion cubic metres), not far off the current US reserve of 153 billion cubic feet.
The looming helium crisis seems to have been averted, for now.
Under pressure
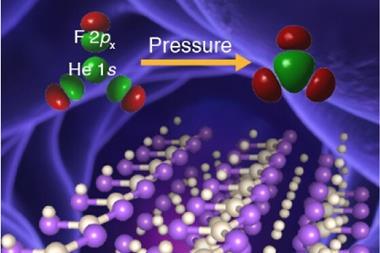
When it comes to chemistry, helium doesn’t leave a lot to report. Helium is the most unreactive element on the periodic table, possessing a record-high ionisation potential, and its electron affinity is essentially zero. While other members of the noble gases have been coaxed into undergoing some chemistry, particularly with very electronegative elements such as fluorine, helium has resisted – until recently.
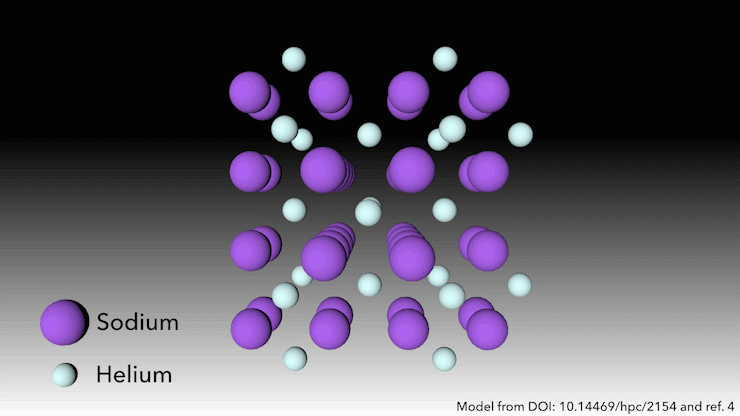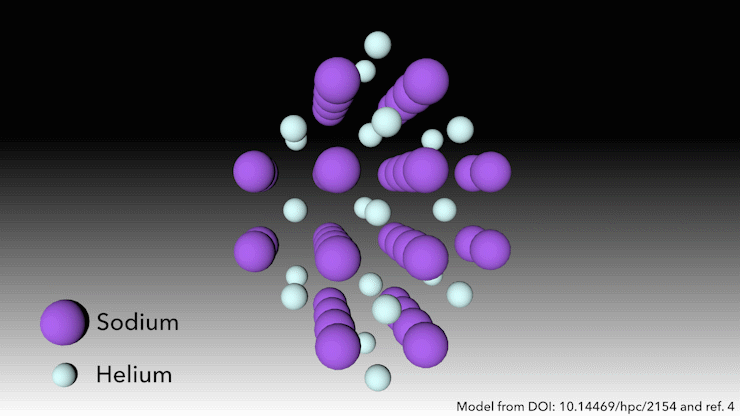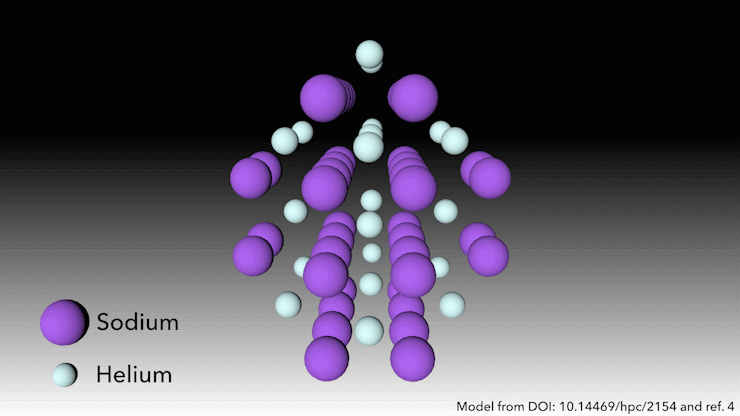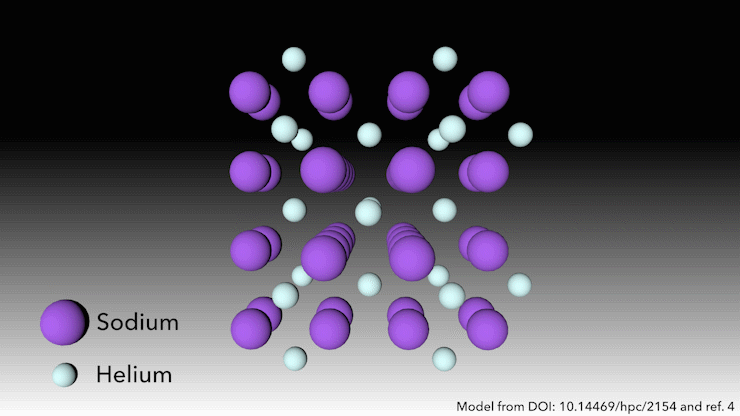
In 2017, an international team showed helium will form a stable solid-state compound when mixed with sodium.4 You just need to squeeze it hard enough. ‘At very high pressures, something weird happens,’ says Richard Dronskowski from Aachen University in Germany, who was part of the team. ‘The helium atom bonds – without really bonding.’
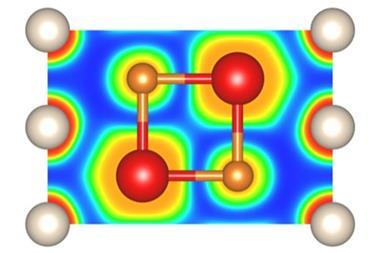
At pressures of over 113 gigapascals – more than one million atmospheres – the sodium atoms in the structure are being squeezed so hard that they spit out their valance electron. ‘The sodium cation is much smaller than the sodium atom – but then the question is, where do we put the electron?’ Dronskowski says. That’s where the helium atoms come in. ‘The helium is the spacer – it doesn’t accept electrons, but it keeps the sodium cation and negatively charged electron away from each other.’ Pairs of electrons sit in holes in the structure that the helium atoms help to create.
This strange helium compound, which has the formula Na2He, is the first example of a solid-state material in which an atom acts as a spacer to help disproportionate an atom into a cation and electron, Dronskowski says. But in other ways, the material is quite familiar, he adds. ‘In a nutshell, it is a salt just like table salt. It adopts a crystal structure that is very well known in solid state chemistry.’
And helium – supposedly totally inert – continues to surprise and lead to new kinds of chemistry. It was computer predictions – much easier to run than expensive and time-consuming high pressure experiments – that first suggested Na2He might exist. New calculations show helium might actually form a whole range of compounds, some of which might be stable at pressures as low as 30 gigapascals.5
Beyond our solar system
Peering out into the cosmos, studying distant chemistry by looking for the tell-tale imprint of spectral lines in light reaching the Earth, is a research topic just as hot today as it was in Janssen and Lockyer’s time.
Exactly 150 years after the two astronomers spotted the bright yellow emission line that was humanity’s first glimpse of helium, a UK-led team of researchers identified helium in the atmosphere of an exoplanet for the first time.6 ‘The process that we used, and that Lockyer used, is actually pretty similar,’ says Jessica Spake from the University of Exeter, who led the research. ‘The yellow line Lockyer found, and the one we found in the near-infrared, both come from excited helium.’

Although helium was widely expected to be present in the atmosphere of planets orbiting distant Suns – as it is within planetary atmospheres of our own solar system – two hurdles had stopped us detecting it, Spake explains. Firstly, there have been few detectors that operate in the near-infrared spectrum. And secondly, water molecules in Earth’s atmosphere have emission lines that obscure the helium signal. ‘It has been a slightly awkward wavelength region to work in,’ Spake says. As a result, helium hunting fell out of fashion.
Spake’s study puts the element back in vogue. Using the near infrared detector on the Hubble Space Telescope, Spake spotted the helium signal as exoplanet WASP-107b passed in front of its star and some of the star’s light passed through the planet’s helium-rich upper atmosphere. That starlight carried helium’s signature emission band all the way to Earth.
‘The exciting thing is, many high resolution spectrographs that do cover this wavelength are coming online in the next year, so now is the perfect time to look for helium,’ says Spake. ‘We think there is quite a lot of science we can do with this new method of studying exoplanet atmospheres,’ she adds.
One of the strangest findings of exoplanet science so far is that the most common type of planet in the galaxy are super-Earths and mini-Neptunes – a type of planet we had no idea existed, as they aren’t found in our own solar system. ‘In our solar system we have small rocky planets and larger ice giants – these exoplanets fall in between,’ says Spake. ‘We don’t know what they are made of.’ One possibility is that they consist of a rocky core surrounded by a large hydrogen and helium envelope. The other leading theory is they are made mostly of water. Measuring their helium signal could help clear up what the most common planets in the galaxy are made of.
And then there’s the possibility of using helium to identifying distant Earths. Simply because of their diminutive size, rocky planets like our own are difficult to pick out in orbit around distant stars. But because the helium emission emanates from planets’ upper atmosphere, the planet appears up to three times bigger at this wavelength than at optical wavelengths, says Spake. Exoplant hunting at this wavelength should make smaller planets easier to spot. The James Webb space telescope, due for launch in May 2020, should be able to pick out Earth-sized exoplanets using this technique.
‘We think helium is hugely important,’ says Spake. ‘We hope it will be used a lot more in coming years.’
James Mitchell Crow is a science writer based in Melbourne, Australia
References
1 H Aldersey-William, Periodic Tales: The Curious Lives of the Elements, Penguin, 2012
2 The Impact of Selling the Federal Helium Reserve, National Academy Press, https://bit.ly/2JkUlEc
3 C Ballantine et al, Goldschmidt conference 2017, https://bit.ly/2NfId5M
4 X Dong et al, Nat. Chem., 2017, 9, 440 (DOI: 10.1038/nchem.2716)
5 Z Liu et al, Nat. Commun., 2018, 9, 951 (DOI: 10.1038/s41467-018-03284-y)
6 J J Spake et al, Nature, 2018, 557, 68 (DOI: 10.1038/s41586-018-0067-5)










1 Reader's comment