Using radioisotopes to image inside patients’ bodies – nuclear medicine – is under threat from ageing reactors. James Mitchell Crow discovers the new science trying to fix the problem
Nuclear medicine is a race against time. From the moment a medical radioisotope is generated, it starts to decay. The half-life of the metastable isotope technetium-99m (Tc-99m), the most commonly used radioisotope for medical imaging, is just 6 hours. Despite this narrow window of use, up to 100,000 medical imaging procedures to diagnose conditions ranging from coronary artery disease to cancer are performed using this short-lived radioisotope worldwide each day.
Recently, however, this essential branch of medicine has been facing another ticking clock: a race against time to shore up the creaking medical radioisotope supply. Most of the small handful of research-scale nuclear reactors that are the world’s source of Tc-99m are reaching the end of their working life. Over the past decade, unplanned shutdowns of ageing reactors have precipitated several critical Tc-99m shortages. Since 2015, two of these venerable reactors, in France and in Canada, have ceased operation altogether. Just six reactors now supply almost all the world’s supply of Tc-99m.
But after a slow start, the race to replace this ageing nuclear infrastructure and to safeguard medical imaging is beginning to look like one we are winning. New and improved supplies of the radionuclide are coming online or are close to completion. Meanwhile, the range of radioisotopes that hospitals use for imaging purposes has diversified, often to isotopes made in synchrotrons rather than nuclear reactors. And on the biochemistry side, powerful new radioisotope-tagged labelling molecules are revolutionising the diagnosis and treatment of conditions ranging from Alzheimer’s to prostate cancer.
You might even say nuclear medicine’s future appears to be glowing.
Radiodiagnostics
Consult a doctor with a mysterious case of chest pains, and you’ll probably be sent for a medical imaging procedure called cardiac perfusion imaging. This test, to assess blood flow to the heart muscle, is one of the most common technetium-99m imaging applications.
‘Technetium is a wonderful nuclide for medical imaging,’ says Jason Holland, who develops radiopharmaceuticals at the University of Zurich in Switzerland. ‘It has a gamma ray emission of 140KeV, exactly in the window you want for high-quality imaging,’ he explains (see box). ‘It is also a generator-based radionuclide, so readily accessible.’

Tc-99m arrives at your hospital’s imaging department inside a toaster-sized technetium-99m generator. It is made in a multi-step procedure, which – appropriately enough for a process pioneered in the 1950s – generally starts with weapons-grade uranium.
This uranium, highly enriched with U-235, is attached to a target and irradiated inside a nuclear reactor. Bombarded by thermal neutrons, the uranium undergoes fission reactions, one of which splits the uranium to give tin-134 and molybdenum-99. Mo-99 decays to Tc-99m with a 66-hour half-life, long enough for a well-oiled logistics system to air-freight it around the globe inside shielded technetium-99m generators.
Within the generator, the Mo-99 is contained on alumina-packed chromatography columns. The radioisotope, adsorbed onto the column in the form of the doubly charged molybdite ions, MoO42–, continually decays to the corresponding TcO4- pertechnate ion. As a singly charged species, TcO4– ions bind less tightly to the alumina and can be selectively eluted from the column by passing standard saline solution through the generator.
Turning the gamma-ray-emitting Tc-99m radioisotope into a useful diagnostic tool involves coordinating it to a molecule that accumulates in a particular part of the body of medical interest. That might be a tumour, or the heart.
To assess a case of chest pains, hospitals typically use the technetium-chelating agents sestamibi or tetrofosmin. These molecules are rapidly and selectively taken up by heart muscle tissue, where they can be imaged by Spect imaging (see box). Once absorbed, the radiotracers can reveal areas of heart tissue that are not receiving enough blood due to blocked or narrowed arteries, guiding the doctor on the next cause of action.
Emission control
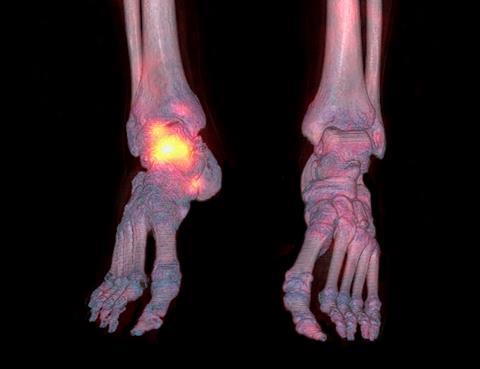
The radioisotopes used for medical imaging decay to release gamma rays – the highest energy, shortest wavelength form of electromagnetic radiation. High-energy gamma rays can easily exit the body and be detected by a camera. The traditional form of emission imaging, using radioisotopes such as Tc-99m, is called single-photon emission computerised tomography (Spect).
‘All image reconstruction using gamma rays relies on the fact you have to draw straight lines,’ explains Matthew Dimmock, a medical imaging researcher at Monash University in Melbourne, Australia. Gamma rays travel in straight lines – so if you can establish that line, you can identify where inside the body the gamma ray was emitted ‘To draw a straight line, you need two points,’ Dimmock says. The camera detecting the gamma ray gives you one point. Spect imaging instruments use a filter pinpricked with tiny holes, called a collimator, to establish the second point on the line to be able to retrace each gamma ray’s path. ‘When you do that many millions of times, the image you build up turns out to be the image of what you see inside the patient,’ Dimmock says.
The downside of Spect imaging is that most of the emitted gamma rays hit the collimator and are blocked by it, limiting sensitivity. That’s not the case with the other emission imaging technique, positron emission tomography (PET), which has dramatically risen in popularity over the past decade or two.
PET isotopes, which include carbon-11 and fluorine-18, decay by emitting a positron from the nucleus. As soon as the positron meets its antiparticle, an electron, they annihilate in a burst of energy – emitting a pair of gamma rays. ‘The two gamma rays are emitted back to back, 180° from each other,’ Dimmock explains. Cameras surrounding the body detect these pairs, giving the two points in space needed to draw the straight line. Without the collimator, PET imaging is up to 1000 times more sensitive than Spect imaging.
Securing the source
Of the six main reactors currently producing Mo-99, four will reach the end of their working lifespan within little over a decade. But others – including the newest, the Opal research reactor opened in Sydney, Australia in 2007 – are expanding their output. Brand new reactors, due to come online within the next five years in France, Germany, South Korea and Argentina, should help pick up the slack.

At the same time, new production technologies are reducing our dependency on nuclear reactors for medical isotopes. Since the permanent closure of Canada’s 60-year-old National Research Universal Reactor at Chalk River in 2018, North America has lacked any Mo-99 production and has had to import all the material its hospitals need. That’s despite the US consuming around half of the world’s supply of this radioisotope. Prompted by shortages in Mo-99 after unexpected Chalk River shutdowns over the past decade – as well as the desire to get away from using weapons-grade uranium in Mo-99 production – US regulators have made a concerted effort to nurture new ways to make it.
Organisation including the US Food and Drug Administration and the US Department of Energy’s Argonne National Laboratory have teamed up with private companies to pioneer these new methods. In February 2018, the FDA approved the first of these, developed by NorthStar Medical Radioisotopes. The company uses neutron capture to convert Mo-98 into Mo-99, using neutrons from US research reactors. The company is also developing a reactor-free version that uses a particle accelerator. High-energy photons generated using an electron accelerator knock out a neutron from naturally occurring Mo-100, transmuting it into Mo-99. A second company, Shine, is developing a reactor-free approach that uses particle accelerators to induce sub-critical fission of low-enriched (non-weapons grade) uranium to give Mo-99.
Supplies of Tc-99m , which still accounts for 80% of nuclear medicine procedures worldwide, look increasingly secure thanks to a diversifying range of sources. Also easing pressure is the fact a zoo of other radioisotopes are now being used for various medical imaging applications, and more are being added all the time. Some radioisotopes complement Tc-99m imaging – others could replace it altogether.
In the complementary camp are the imaging molecules that incorporate a carbon-11 or a fluorine-18 radioisotope. These isotopes can be imaged using positron emission tomography (PET). The great advantage of these radioisotopes is that they can be incorporated into small organic molecules that can cross the blood–brain barrier and therefore light up particular structures in the brain. ‘It is a growing area,’ says Neil Vasdev, associate director and chief radiochemist of the research imaging centre at the Centre for Addiction and Mental Health in Toronto, Canada.
Applications to Alzheimer’s
One area of major medical interest that PET imaging is helping to illuminate is in neurodegenerative diseases such as Alzheimer’s disease. Despite decades of research, Alzheimer’s lacks any treatment that can slow the progression of the disease.
Until recently, developing Alzheimer’s drugs was complicated by the fact the disease couldn’t even be definitively diagnosed in living patients. The characteristic brain damage – shrunken brain structures clogged up with clumps of two proteins, amyloid and tau – could only be identified post mortem. But around 15 years ago, researchers William Klunk and Chester Mathis at the University of Pittsburgh in the US changed that. They created a molecule based on thioflavin T, used as a histological dye for amyloid imaging of brain tissue, into which they incorporated a C-11 atom. Known as Pittsburgh Compound B (PiB), it lights up amyloid plaques in PET scans. ‘Plaque imaging has really revolutionised the field,’ Vasdev says.
We are very limited in how we label molecules and drugs with carbon-11 and fluorine-18
PiB’s downside is that C-11’s half-life is just 20 minutes, which meant each batch could only be used on a couple of patients at most. Several amyloid-labelling PET radiotracers that use F-18, with its 2-hour half-life, have since been developed. Alzheimer’s clinical trials are currently underway that use PET imaging to identify patients before they show any symptoms, in the hope that early intervention with amyloid-busting drugs will stave off the disease.
But radiochemists like Vasdev aren’t resting on their laurels. ‘One of the biggest thrusts of our research is to look at PET imaging agents beyond amyloid,’ Vasdev says. ‘We have over 10 approaches for imaging Alzheimer’s that we are trying, to look at the different hypotheses of Alzheimer’s.’ One possibility is that amyloid is just a bystander in the disease, and that the tau protein is the real culprit of neurodegeneration, so the team is working on PET imaging agents for tau, for example.
A big part of the challenge Vasdev faces is developing the organic chemistry to incorporate a C-11 or F-18 atom into a tracer molecule. The short half-lives of these radionuclides demand that they are added at the last, or penultimate, step in the synthesis.
‘We are very limited in how we label molecules and drugs with carbon-11 and fluorine-18,’ Vasdev says. ‘With F-18 it is almost always nucleophilic displacement with fluoride ions. With C-11, the norm is to do methylation with C-11 labelled methyl iodide or methyl triflate. Our lab is working to push the limits of what we can do.’
With metal ions, the advantage is flexibility of chemistry
One radiolabelling method Vesdev and his team has helped implement is a copper-catalysed method for C-11 carbon dioxide fixation, generating molecules incorporating a C-11 labelled carboxylic acid.1 Another fruitful area Vasdev and his team has developed is a method to readily introduce F-18 to non-activated aromatic rings. Vasdev’s improved method starts with the corresponding iodide of the non-activated aromatic ring they wish to radiofluorinate. They convert the iodine to a stabilised hypervalent iodinium ylide, which is readily turned into the desired radiofluorinated compound by reacting it with fluoride-18 ions.2
The team recently used this strategy to make an F-18 radiotracer to investigate metabotropic glutamate receptor 5 (mGluR5), which is known to be upregulated in early Alzheimer’s disease. Other projects include developing radiotracers to investigate receptors associated with opioid addiction and mood disorders.
In the blood
Although the blood–brain barrier imposes limitations on the radionuclides that can be used for brain imaging, for the rest of the body there is no such restriction. A number of metals have isotopes that can be imaged with PET, including copper-64 and gallium-68.
‘With metal ions, the advantage is flexibility of chemistry,’ says Holland. ‘You have the ability to coordinate different ions that have different chemistry and different radioactive properties, and couple them to a very wide array of different molecules.’
Metals are proving to be particularly useful for PET imaging of tumours. The set-up typically starts with a molecule such as a peptide or antibody that tightly binds an antigen or receptor found in high concentration on the surface of tumour cells. To turn that molecule into an imaging agent, it is conjugated to a chelating group that binds the radioactive metal. Holland and his team are currently focused on developing new imaging agents for prostate cancer.
‘We are trying to come up with new chelates to allow us to coordinate different metal ions in a very rapid and efficient way,’ says Holland. ‘We are also looking at new ways to take our metal-binding chelates and couple them in a specific way to different proteins.’
Search and destroy
Synthetic inorganic chemist Paul Donnelly at the University of Melbourne in Australia is trying to take nuclear medicine beyond diagnosis and into treatment. Donnelly has struck up a productive partnership with Rodney Hicks, who directs the centre for cancer imaging at the Peter MacCallum Cancer Centre in Melbourne. The pair is particularly interested in taking diagnostic radiopharmaceuticals that light up tumours and, by switching the radionuclide, turning them into highly targeted therapeutic agents that deliver radiotherapy right to the tumour.
Radionuclides that are useful for imaging emit gamma rays, which largely pass through the tissues and exit the body. Radionuclides with therapeutic potential for treating tumours decay by emitting alpha or beta particles, which only travel a short distance before they strike a nearby molecule. In cancer therapy, the emitted particles kill tumour cells by causing double-strand breaks in their DNA. ‘The particles’ path length is one to two millimetres, so we can give a very high dose with minimal collateral damage [to surrounding healthy tissues],’ Hicks explains.
So far, Hicks has mainly concentrated on pairing diagnostic gallium-68 with therapeutic lutetium-177 – but it is a combination with a couple of downsides, he explains. ‘When you label a biomolecule, the bigger the radionuclide the more likely you are to change its biological characteristics, including the affinity for the receptor,’ Hicks says. ‘Gallium-68 and lutecium-177 have similarities, but are not the same metal.’ The other drawback is gallium-68’s 68-minute half-life. ‘We can only model the very early stages of the uptake of the biomolecule in cells, we can’t really ascertain what the retention of that molecule in the tumour is going to be.’
A much better diagnostic–therapeutic pairing would be to use a couple of copper radionuclides. Copper-64 is already used for PET imaging, and its 12.7-hour half-life is long enough to image uptake and subsequent clearance of a radiopharmaceutical. Copper-67, meanwhile, is a beta-particle emitter with excellent therapeutic potential, says Hicks. As the in vivo behaviour of the two labelled radiopharmaceuticals should be identical, using PET imaging to track uptake and clearance of the former would allow very precise dosing of the latter, a process known as quantitative prospective radiation dosimetry.
New chemistry developed by Donnelly is crucial to this work, says Hicks.3 Previous copper-chelating agents did not quite bind the copper strongly enough, and would slowly leak the metal. ‘Previous attempts were doomed to failure because if the Cu-67 falls off, it is giving radiation to normal tissue and not the tumour any more,’ Hicks says.
Donnelly took a known copper-chelating agent, a macrobicyclic hexaamine cage ligand known as a sarcophagine, and modified it – in particular, developing a pendant carboxylic acid group that could be reliably attached to an amine on a tumour-targeting peptide Hicks was working on. The peptide binds somatostatin receptors overexpressed in many types of neuroendocrine tumour. ‘I worked on the chemistry for at least five years before we came to Rod to present him the idea,’ Donnelly says. Hicks immediately recognised the potential. After successful preclinical testing, the team is now conducting first-in-human trials.
A huge amount of cost goes into dealing with the waste from nuclear reactors
Meanwhile, Donnelly and his team are looking at other targeting biomolecules to tag with copper radionuclides. ‘We have a linker and chelator, and we have been working on ways to attach that to whatever biomolecule you want,’ Donnelly says. Projects include an antibody that targets the HER2 receptor associated with some breast cancer cases. Targeting the prostate specific membrane antigen to tackle prostate cancer is another potential avenue for copper theranostic radiochemistry, Hicks adds.
At the moment, Cu-67 supplies are limited and the team has to get the radionuclide from a linear accelerator at Idaho State University in the US. ‘People say “You can’t get Cu-67, so why develop a radiotheranostic agent for that?”’ says Hicks. ‘But when I started doing lutetium therapy there was only one reliable supplier, and now there’s a whole heap of them.’ There is no reason why the same can’t happen with Cu-67, he says. ‘If the therapeutic trials we are doing are positive and we demonstrate the utility of this technique, there will be a greater incentive to industrialise the Cu-67 production process.’ There is no reason other particle accelerators around the world couldn’t start to produce it, he adds.
In fact, in terms of diagnostics, there’s no reason we couldn’t switch all procedures to radionuclides produced by particle accelerators rather than in nuclear reactors, Hicks argues.4 ‘A huge amount of cost goes into dealing with the waste from nuclear reactors, plus there are the safety concerns, the environmental concerns.’ By comparison, radionuclides produced by cyclotrons are extremely cheap and have no waste products in the vast majority of cases. ‘It is a no-brainer they should be used,’ says Hicks. ‘To me, it should be inevitable.’
James Mitchell Crow is a science writer based in Melbourne, Australia
References
1 B H Rotstein et al, ACS Med. Chem. Lett., 2014, 5, 668 (DOI: 10.1021/ml500065q)
2 B H Rotstein et al, Nat. Commun., 2014, 5, 4365 (DOI: 10.1038/ncomms5365)
3 B M Paterson et al, Dalton Trans., 2014, 43, 1386 (DOI: 10.1039/c3dt52647j)
4 R J Hicks and M S Hofman, Nat. Rev. Clin. Oncol., 2012, 9, 712 (DOI: 10.1038/nrclinonc.2012.188)













No comments yet