Inorganic chemistry – Page 21
-
 News
NewsLive blog: New element names
The proposed names of the four new elements have been released. Find out what they are in our live coverage.
-
 Research
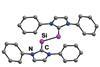
ResearchRare mixed-valent Si2H radical isolated
N-heterocyclic carbenes used to trap subvalent disilicon hydride species
-
 Podcast
PodcastTetraethyl lead
For decades, automobile fuel had a dangerous additive: Tetraethyl lead. Matt Gunther explains why a toxic compound was added to our fuel
-
 Research
ResearchAll-metal sandwich inspires a theoretical following
Recently discovered [Sb3Au3Sb3]3– has multiple groups hunting for answers
-
 Podcast
PodcastUranium dioxide
This week’s compound has had a glowing career in the arts and a runaway success in the energy sector
-
 Business
BusinessQuotient ups carbon-14 quota with recycling plant
Plant may provide a solution to the supply shortage of the radiolabel barium [C-14] carbonate
-

-
 News
NewsBeyond element 118: the next row of the periodic table
A technological leap may be called for to expand the number of elements in existence
-
 Research
ResearchElectron work functions look tough
Surface property calculations provide a link between the electron work function and toughness of transition metals
-
 News
NewsConfirmation of four new elements completes seventh row of periodic table
Iupac endorse discovery of elements 113, 115, 117 and 118 by teams in Russia, Japan and the US
-
 Feature
FeatureDating the age of humans
Physical science is helping archaeologists close in on the real answers behind the mysteries of human evolution, finds Ida Emilie Steinmark
-

-
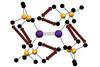 Research
ResearchSubtle forces yield profound effects on heavy element bonding
Calculations on dispersion forces shed light on interactions between heavier main group elements
-
 Podcast
PodcastChemistry World podcast – May 2015
We find out how nanotoxicology could be holding back development, and ask if ‘patent or perish’ should be the new academic adage
-
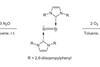 Research
ResearchLonger-lived oxides offer silicon synthesis boost
Touted as ‘soluble sand’, stabilised small silicon oxides present new synthetic worlds
-
 Research
ResearchLawrencium experiment could shake up periodic table
Measurement of first ionisation energy confirms electronic configuration but opens up an important debate
-

-

-
 Feature
FeatureGoodenough rules
Bea Perks profiles a veteran scientist whose fundamental and applied research continues to shape the world we live in
-
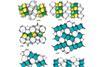 Research
ResearchZeolites net new carbon allotropes
Six new forms of carbon predicted using known topologies from the zeolite field