A fullerene-like molecule composed entirely of metal atoms has been made by researchers in China. As the molecule is highly unstable, it remains uncertain whether it will find direct applications, but its existence could provide new insights into the bonding between metal atoms.
The football-shaped carbon allotrope buckminsterfullerene (C60), whose unexpected 1985 discovery won Harry Kroto, Robert Curl and Richard Smalley a Nobel prize, has gone on to find use in impact-resistant lightweight materials, solar cells and even medicinal applications. Analogous inorganic fullerene structures have since been produced from compounds such as tungsten disulfide and molybdenum diselenide. But purely metallic fullerenes have remained elusive: theoretical calculations predict that a spherical Au32 form of gold should exist, but – perhaps because of the difficulty of characterising x-ray structures in the gas phase – it has only been detected when stabilised by external ligands.
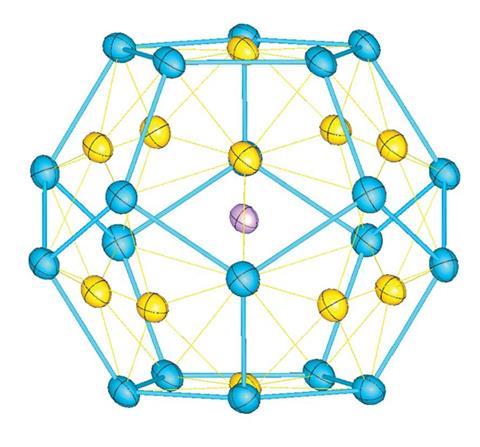
In the new work, Zhong-Ming Sun at Nankai University in China and colleagues crystallised a compound in which the potassium–gold–antimony [K@Au12Sb20]5- anion is a dodecahedral cluster. A gold atom sits at the centre of each face, with an antimony atom at each vertex. At the centre is a potassium atom, which templates the structure by coordinating to all 12 gold atoms.
Like the original fullerene discovery, the Chinese researchers’ findings were unplanned: ‘Before we started the experiment we had no idea what would be formed,’ says Sun. ‘We thought it should be something new but really nobody knew what would happen.’
The molecule’s instability may preclude applications, but Sun says the insights it provides into bonding between metal atoms could prove invaluable. ‘Nobody thought about forming this kind of structure, but the butterfly shape of the two gold and the two antimony atoms forms a four-centre, two-electron bond,’ says Sun; ‘This structure can be considered as a kind of super atom because it’s spherical aromatic – and this is kind of new.’
‘It’s a beautiful [study] on the structure of a new metallic molecule with dodecahedral symmetry,’ says physical organic chemist Harry Dorn at Virginia Tech; ‘I have a feeling it’s kind of a stretch to relate it back to a fullerene, because fullerenes can entrap five, six or seven atoms inside.’
Dorn is concerned that the molecule was too unstable for the researchers even to obtain a mass spectrum, but be believes similar molecules might be stabilised in future. He finds the formation mechanism especially interesting: ‘I haven’t seen anything like that where you go from things that have low symmetry to things that have as high symmetry as this does,’ he says. ‘I come back to the beauty of it.’
References
Y-H Xu et al, Science, 2023, DOI: 10.1126/science.adj6491














No comments yet