Gene editing technique is developing apace with two new technologies unveiled
The therapeutic potential of the Crispr/Cas genome editing tool continues to grow, as US scientists have developed a version of the system that targets RNA and chemically alters its nucleotides.1
As RNA is ultimately translated to make proteins, being able to edit it could be advantageous for research and gene therapy applications, says Feng Zhang who led the team behind the research at the Broad Institute and Massachusetts Institute of Technology in the US.
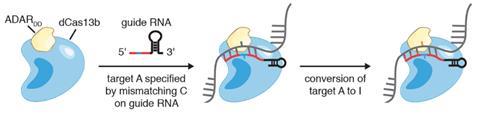
‘Many genes can play different roles depending on when and where they are expressed, and by modulating them at the level of RNA, we can begin to tease apart these different roles,’ he says. ‘RNA editing also has great potential as a therapeutic because its administration can be regulated and dosed.’
The system Zhang and colleagues developed uses a bacterial enzyme called Cas-13 to hone in on messenger RNA (mRNA) within cells, much like the way Cas-9 is used to identify and cut DNA. However, instead of using the Cas-13 itself to alter the RNA they created a ‘dead’ version that could still target RNA but was catalytically inactive. They fused this to an adenosine deaminase enzyme, which is capable of converting the A (adenosine) nucleobase to inosine, which is read by cells as a G (guanosine).
By combining this with a guide RNA that is complementary to the target site, they designed a system – which they called RNA editing for programmable A to I replacement, or Repair – that can target a specific mRNA transcript.
‘The guide RNA is specially designed to have one mismatch in to the target site, opposite the exact adenosine to be edited,’ explains Zhang. ‘This mismatch creates a bubble for [the adenosine deaminase] to work in, allowing for precise editing of the target A site.’
‘Repair can achieve precise, efficient single-base changes within an RNA transcript,’ he adds. ‘[It is] like using the “find and replace” function in a word processor to correct typos.’
Helen O’Neill, a molecular geneticist at University College London in the UK, says the technique ‘paves the way for selectable and time specific modulation of a gene’s output’. ‘Manipulating RNA potentially poses fewer ethical concerns than editing DNA as the effects would not necessarily be permanent,’ she adds.
In a separate study also published earlier this week, another group at the Broad Institute used Crispr to edit individual DNA base pairs within bacterial and human cells. The system – which uses an adenosine deaminase coupled to an inactive Cas-9 – can convert adenosine–thymine (A-T) complementary pairs to cytosine–guanine (C-G) ones with around 50% efficiency.2 Previous Crispr technologies could only change C-G to A-T or snip out and swap in larger pieces of DNA. Although applications in clinical gene therapy are still a long way off, the researchers point out that the approach shows promise, given that the mutation of a single C-G base pair into a rogue A-T one is involved in several genetic diseases.
‘This has been an exciting week for genetic research,’ says O’Neill. ‘These papers highlight the fast pace of the field and the continuous improvements being made in genome editing, bringing it closer and closer to the clinic.’
References
1 D B T Cox et al, Science, 2017, DOI: 10.1126/science.aaq0180
2 N M Gaudelli et al, Nature, 2017, DOI: 10.1038/nature24644















No comments yet