Two researchers in the US have developed a simple, cheap and effective method for recycling catalytic organoiridium waste into a synthetically important iridium precursor. This is the first lab-scale example of such a recycling process, which could be a practical and cost-effective option for many of the academic research groups working with this rare element.
‘Precious metal waste from academic labs used to be collected separately and returned to the supplier who would recycle these concentrated waste streams and repay us with fresh starting materials,’ explains Ulrich Hintermair, whose research at the University of Bath in the UK involves catalysis. ‘These recycling schemes ceased about 10 years ago … it’s become too costly to run these recycling schemes safely, so we now just dispose of all these precious elements like any other laboratory waste, which gets incinerated in large facilities. Madness from a sustainability point of view.’
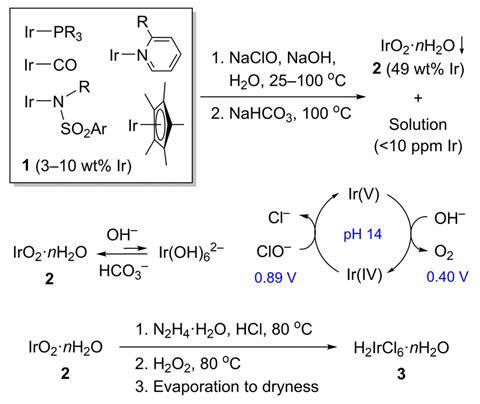
Now, Travis Williams and Valeriy Cherepakhin at the University of Southern California have put forward an alternative method for dealing with organoiridium waste. Their method uses sodium hypochlorite (bleach) alongside other cheap reagents, such as sodium hydroxide and sodium bicarbonate to make hexachloroiridic acid. ‘With other precious metals, [recycling industries] are trying to go to an ore… we knew we wanted to go to a chloroiridate because that’s the [starting point] from which all commercial iridium complexes are made,’ says Williams.

The process begins by oxidising the organoiridium waste in sodium hydroxide solution before briefly heating it with bleach to generate an iridium hydroxide intermediate. This precipitates out of solution using excess sodium bicarbonate at 100°C before they re-dissolve it in hydrochloric acid, in the presence of hydrazine. Adding hydrogen peroxide to the solution oxidises any remaining Ir(III) and destroys any excess hydrazine, before the solution is evaporated down to provide hexachloroiridic acid, which Williams and Cherepakhin claim can be used without further purification. To demonstrate this, they used the resulting hexachloroiridic acid as a starting material to synthesise [(1,5-cyclooctadiene)IrCl]2 with a yield of 87%.
Catalysis experts are excited by the idea. ‘The price of precious metals has been going up so there is a big incentive for researchers to recover iridium, especially if it can be done in a simple and safe manner. I expect there will be a lot of interest in this work… I am eager to test out their procedures in my own lab,’ comments Loi Do from the University of Houston in the US. And Andreas Pfaltz, from the University of Basel in Switzerland says ‘for research groups who use iridium and acquire a lot of iridium waste, this presents a good solution for them. It looks like a very good solution to solve a niche problem, and they’ve solved it well.’
References
T Williams and V Cherepakhin, Green Chem., 2024, 26, 3146 (DOI: 10.1039/d4gc00151f)












No comments yet