Scientists in Japan have used carbon nanobelts in a Diels–Alder reaction with alkynes and arynes to form the largest iptycene to date.
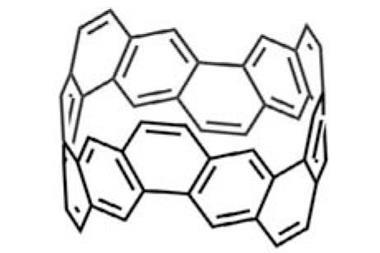
Carbon nanobelts are a short cyclic system made of fused benzene rings. Despite being predicted in 1954, they were only synthesised for the first time by Kenichiro Itami’s team a few years ago.
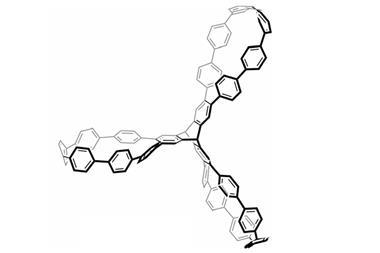
Now, Itami and his team at Nagoya University have discovered that (6,6)carbon nanobelts can react along the central benzene rings in a six-fold Diels-Alder reaction to form cycloiptycene derivatives in one step. The method created cyclotetracosiptycene, the largest iptycene reported since nonadeciptycene in 1990. The (6,6)carbon nanobelts showed reactivity towards different alkynes and arynes to obtain a range of unusual, highly symmetric iptycene derivatives. Although closely related nanotubes have indicated how carbon nanobelts might react, these are the first reported experiments to explore their reactivity.
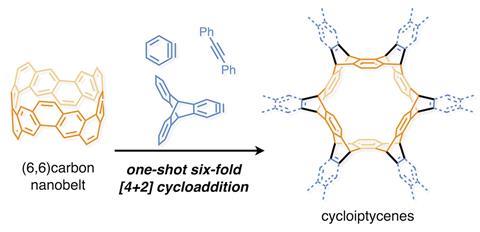
Iptycenes consist of arenes separated by a bridgehead system around a bicyclo-octatriene core and are challenging to synthesise. To form cyclotetracosipticene, the team heated a triptycene-based aryne with (6,6)carbon nanobelts and caesium fluoride in acetonitrile and 1,2-dichloroethane at 85°C for seven days. Although they could only isolate 5% of the product, x-ray crystallography confirmed its structure.
Such sidewall reactivity shows that carbon nanobelts could be versatile building blocks for large macrocyclic carbon-based structures.



















No comments yet