Chemists have revealed a gas-phase nucleophilic substitution reaction where the molecule first ‘flips over’ before the nucleophile attacks, leaving the molecule’s configuration unchanged. ‘These [substitution reactions] are quite complicated and maybe these are more complicated than we learn,’ says theoretical chemist Gábor Czakó at the University of Szeged in Hungary, who was not involved in the work.
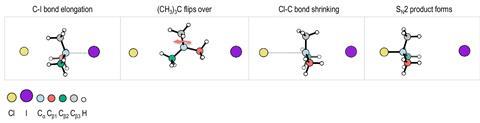
Bimolecular nucleophilic substitution – commonly known as SN2 – is one of the most fundamental reactions in chemistry. Chemists often describe the reaction’s mechanism as a nucleophile attacking the back face of a central carbon atom, followed by the removal of a leaving group. This Walden inversion mechanism switches the configuration of the tetrahedral carbon.
‘This is for SN2 [reactions] in the lowest transition state,’ says Roland Wester at the University of Innsbruck in Austria. ‘But then there is also this slightly higher channel where the molecule just behaves differently.’
Chemists have previously discovered other substitution reactions that do not lead to inversion, including ‘double-inversion’ or ‘front-side attack’ mechanisms.
Wester’s team, along with researchers at the Dalian Institute of Chemical Physics in China, has now uncovered another mechanism where gaseous tert-butyl iodide reacts with chloride ions via a ‘flip-over’ mechanism.
‘The nucleophile comes in, but then instead of [the normal inversion mechanism], the molecule flips over,’ explains Wester. Elongation of the carbon–iodine bond allows the molecule to reconfigure before the chloride nucleophile attacks on the same face as the iodine leaving group. Wester says that this retains the original configuration of the molecule.
Firing gaseous chloride ions at an angle of 60° to a beam of tert-butyl iodine molecules allowed the team to study individual substitution reactions. Measuring the velocity and direction of ejected iodide ions gave information about how the substitution had taken place.
The team conducted computational studies of such collisions using a potential energy surface with 39 dimensions – the tert-butyl iodine’s 45 total degrees of freedom minus three translational and three rotational modes. ‘The potential energy surface is based on energies obtained by density functional theory, which is not the highest quality but for this size of system, it’s the best one can do,’ explains Czakó.
Repeating both experiments for thousands of collisions revealed the new ‘flip-over’ mechanism was taking place in about 1% of all substitution reactions.
Doubling the collision energy decreased the probability of an SN2 reaction taking place and bimolecular elimination became increasingly prominent. However, the proportion of substitution reactions that went via a ‘flip-over’ mechanism increased from 1% to 7%, as the increased energy was able to overcome the activation barrier needed for this type of substitution.
‘These collision energies are high above what solution phase chemists are used to,’ says Wester, adding that ‘it’s unlikely that something similar happens in [liquids]’.
Going forward, the team hopes to study more complex molecules to see whether this flip-over mechanism still occurs. Wester explains that exploring the effect of solvation on the reaction by encapsulating gaseous nucleophile ions with water molecules is another goal.
‘It’s interesting to see that a reaction that everybody believes or learns is an inversion reaction also has a mechanism where inversion doesn’t take place,’ says Wester. He adds that this shows that ‘things are hardly ever as simple as they seem’.
References
X Lu et al, Nat. Commun., 2026, 17, 3947 (DOI: 10.1038/s41467-026-72121-4)















No comments yet