For decades, chemists have viewed the [1,2]-Wittig rearrangement as a stubborn rule-breaker – a powerful carbon–carbon bond-forming reaction believed to proceed through unruly radical pairs and largely beyond precise catalytic control. But a new study shows it can in fact be steered with accuracy, opening a route to enantioenriched tertiary alcohols.
The Wittig rearrangement belongs to a broader class of reactions known as sigmatropic rearrangements, which are atom-economic processes that reorganise bonds through well-defined transition states and are widely used to build complex molecules.
Among them, the [2,3]- and [1,2]-variants are especially important. Yet while [2,3]-rearrangements are relatively ‘well behaved’, the [1,2] version has long resisted stereochemical control. ‘[1,2]-rearrangements are forbidden to occur in a concerted fashion … making enantioselective versions rare and particularly challenging,’ explains Andrew Smith at the University of St Andrews.
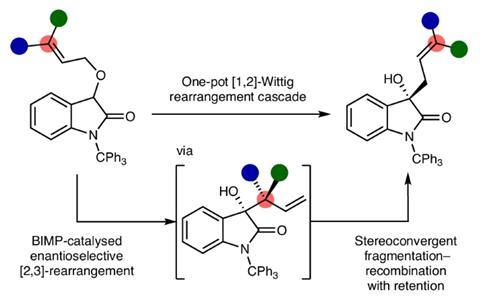
Smith, Matt Grayson at the University of Bath and their teams have uncovered a highly enantioselective route to [1,2]-Wittig products that sidestep the long-accepted radical mechanism entirely. ‘[At the beginning, we] had a simple hypothesis: adding steric bulk at the terminus of an alkene would disfavour [2,3]-rearrangement and lead to [1,2]-rearrangement,’ says Smith. ‘Looking back, we couldn’t have been further from the truth and were amazed at the pathway we uncovered.’
Unlike the better-known Wittig olefination, which forms alkenes, the Wittig rearrangement shifts a group from oxygen to carbon and often generates a tertiary alcohol with a new chiral centre. Because the reaction typically proceeds through highly reactive intermediates, controlling its three-dimensional outcome has historically been difficult.
The new catalytic approach therefore turns a once unpredictable rearrangement into a reliable method for setting stereochemistry while forming a carbon–carbon bond. ‘To fully understand the process, we relied upon computational analysis, and even that threw up surprises,’ says Smith.
‘Most of the experimental data the team accumulated could have been consistent with either a radical or anionic pathway for the fragmentation and recombination event. The single most definitive evidence came from our computational analysis, which did not find any evidence for a radical pathway,’ adds Grayson.
The transformation proceeds through an elegant reaction cascade: a chiral bifunctional iminophosphorane catalyst first guides a precise [2,3]-sigmatropic rearrangement, setting the molecule’s handedness. This is followed by a base-driven fragmentation–recombination step that transfers chirality with remarkable fidelity, ultimately delivering the coveted [1,2]-Wittig products in up to 97:3 enantiomeric ratio.
‘The authors provide strong mechanistic evidence supporting their proposal,’ says Fernanda Duarte at the University of Oxford who was not involved in the study. ‘This work goes beyond a workaround. It represents a conceptual expansion of chemists’ understanding of rearrangement pathways, especially when considering stereochemistry.’
Duarte argues that a fuller understanding of these rearrangements requires better integration of complementary approaches, including synthesis, kinetics and computational methods, as well as the expert judgement needed to connect them. ‘Rearrangements are always full of surprises, and their understanding requires careful analysis,’ she says. ‘In an era increasingly driven by machine learning approaches, this paper highlights that classical physical–organic reasoning and mechanistic tools remain indispensable. These “outliers” [and] unexpected outcomes are the ones that give us access to new areas of chemical space and new products.’
References
T Kang et al, Nat. Chem., 2026, DOI: 10.1038/s41557-025-02022-4












No comments yet