A new analytical method has enabled continuous real-time monitoring of the biosynthesis of sialic acid, an important component of cell surfaces, via NMR. Described as a magnifying glass by lead researcher Christian Hackenberger from the Leibniz Research Institute for Molecular Pharmacology in Germany, the NMR technique allowed them to observe individual enzymatic steps and, for the first time, directly measure their conversion velocities in situ.
Sialic acids are typically found on the termini of glycans, which decorate the surfaces of cells and mediate myriad cell interactions. Metabolic glycan engineering is a popular method that exploits glycobiological pathways to introduce specific chemical entities into cells that are often later functionalised with biorthogonal handles. Due to the promiscuity of the enzymes involved in sialic acid biosynthesis, many metabolic glycan engineering methods use derivatives of N-acetylmannosamine, one of the first carbohydrate species in the pathway. Until now, scientists believed that N-acetylmannosamine kinase processes external derivatives of N-acetylmannosamine.
While many carbohydrate pathways have been studied for years, numerous questions remain regarding their intricacies. As Christian Büll, a glycobiology researcher at Radboud University in the Netherlands, put it, ‘we feed some synthetic sugars like mannosamine-based sugars to cells, and we just look at the glycan output, but what happens in between is still quite a black box.’
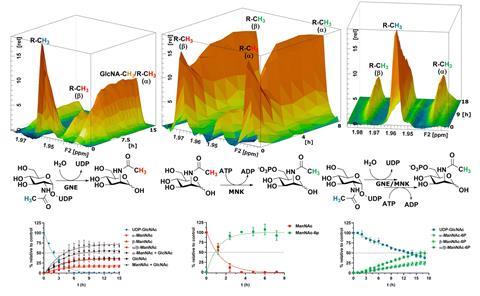
The research team, led by Hackenberger, was able to differentiate between forming monosaccharide species in situ, both using recombinant enzymes and in cytosolic rat liver extracts. They employed state-of-the-art NMR techniques, including pseudo 2D and 3D NMR, to follow minute differences in the characteristic shift of the N-acetyl methyl group signal in the biosynthesis intermediates and directly measured the enzymatic conversion velocities for the first time. Using these methods, they demonstrated that external N-acetylmannosamine is likely introduced into the sialic acid pathway via phosphorylation by N-acetylglucosamine kinase rather than N-acetylmannosamine kinase. ‘There’s been a long debate on which kind of enzymes really tolerate these unnatural mannosamine derivatives and these substrates,’ Hackenberger explains. ‘With this assay, we were, in the end, able to test some of the enzymes which have been speculated to be responsible for some of these additional processes, and we had this really remarkable finding.’
This NMR technique is notable for its ability to monitor the enzymatic processes in a more natural and complex environment, unlike traditional kinetic measurements done in isolation. Hackenberger explains that it ‘allows you to really monitor the individual steps of the biosynthesis in a system which is very close to what happens in nature.’ Martin Tanner, an expert in mechanistic enzymology at the University of British Columbia in Canada, echoes this sentiment: ‘They were able to follow these reactions in a cellular extract which might be tricky to do because you’re taking sort of a garbagy mixture out of a cell and trying to do NMR.’ Although more difficult, he says that more can be learned by looking at the ‘reality of a biological system as opposed to just putting enzymes in test tubes.’
By facilitating insights into sialic acid and other carbohydrate biosynthesis processes, this assay could enhance research techniques like metabolic glycan engineering and potentially medical treatment for disorders of these enzymes. The finding that external N-acetylmannosamine is likely phosphorylated by N-acetylglucosamine kinase has implications for treatment options for GNE myopathy, a hereditary inclusion body myopathy linked to the sialic acid biosynthesis pathway. ‘Any kind of insight which could lead to the understanding of the performance of [GNE] is, of course, central to advancing pharmacological approaches to treat this disease,’ says Hackenberger.
References
This article is open access
J L Gorenflos López et al, Chem. Sci., 2023, 14, 3482 (DOI: 10.1039/d2sc06986e)












No comments yet