A newly reported copper(I)-catalysed allene–ketone addition (CuAKA) offers something rare: a click reaction that forms a robust yet reversible carbon–carbon bond under biologically relevant conditions.
Click chemistry has built its reputation on reliability – fast, selective reactions that work even in messy biological environments. But the permanent bonds it creates can limit applications such as drug delivery and responsive biomaterials, where connections may need to come apart on demand.
‘It is somewhat ironic that the original impetus for click chemistry was the interest in designing and developing functional molecules and yet we were told to aim for really stable linkages, namely those that are devoid of function,’ says Amir Hoveyda at the University of Strasbourg and Boston College. ‘We challenge two general assumptions. [One,] C–C bond forming reactions are not suitable for click chemistry, [and two,] the best click reaction is one that is strongly favored thermodynamically – in other words, it makes an indestructible linkage.’
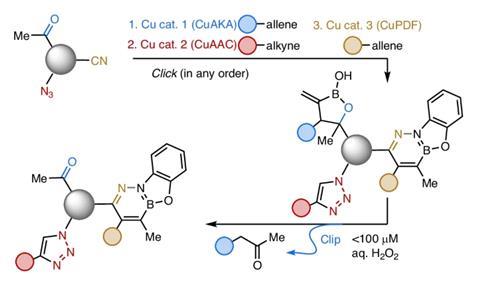
Carbon–carbon bond formation, particularly via carbonyl addition, has long been considered incompatible with click chemistry’s stringent requirements for selectivity, simplicity and orthogonality. But CuAKA proceeds smoothly in aqueous media and tolerates complex biomolecules, allowing direct coupling of drug-like fragments, such as anticancer candidate camptothecin, to cell penetrating peptides.
‘The two-step sequential reaction is … exciting and interesting,’ comments Yimon Aye at the University of Oxford, who was not involved in the study. ‘The first step, involving π-bond breaking and C–C bond making [with a] carbonyl, is unexpected compared to existing click coupling, and could potentially open up new click coupling avenues.’
The resulting linkage can be selectively cleaved at physiological temperature at low hydrogen peroxide concentrations. Equally important is the reaction’s orthogonality. CuAKA operates alongside established copper-catalysed click processes, including CuAAC and CuPDF, without cross-reactivity, allowing multiple click reactions to be combined within a single molecular system.
Still, translating the chemistry into biological settings may prove challenging. Aye notes that naturally occurring carbonyl groups in cells could complicate selective labelling, while the hydrogen peroxide required for cleavage has diverse biological signalling roles and can be difficult to control spatially. However, she adds that local differences in peroxide concentrations might eventually be exploited for targeted cargo release in specific cellular environments.
‘For use in functional biological contexts,’ Aye says, ‘rigorous road-testing and validations in biological systems for each step of the two-step process would be necessary.’
The implications are also potentially broad. In drug delivery, CuAKA could enable conjugates that remain intact during circulation but release their payload in oxidative environments, such as inflamed or cancerous tissue. In chemical biology, it offers a way to install and later remove probes or labels with temporal precision. And in materials science, it opens the door to responsive polymers and networks that can be assembled and disassembled under mild conditions.
‘The catalyst needed is simple, cheap and easy to handle,’ says Hoveyda. ‘The transformation is robust, proceeds at ambient temperature within just a few hours, without needing rigorous control of air or moisture.’
By demonstrating that even traditionally ‘forbidden’ bond constructions can meet click criteria, Hoveyda suggests that this toolbox is far from complete.
References
M E Hackey et al, Nat. Chem., 2026, DOI: 10.1038/s41557-026-02100-1













No comments yet