Chris Smith
This week, flowers, fibre optics and the element that can't quite make up its mind whether it's a metal or not. Taking us back to school, here's Brian Clegg.
Brian Clegg
If there were a competition for the chemical element mostly likely to generate schoolboy howlers, the winner should be germanium. It's inevitable that the substance with atomic number 32 is quite often described as a flowering plant with the common name cranesbill. Just one letter differentiates the flower geranium from the element germanium – an easy enough mistake.

We know germanium isn't a flower, but it's slightly harder to say just what it is. Most elements are either metals or nonmetals. Germanium falls in the same group as carbon and silicon, but also as tin and lead. Germanium itself is classified as a metalloid. It's hard at room temperature and looks metallic with a shiny silvery grey finish, but it's a semiconductor, without some of the key properties of a metal.
Germanium's existence was predicted before anyone isolated it. This was a triumph for Dmitri Mendeleev in his construction of the periodic table. By 1869, Mendeleev had assembled a crude table of the known elements, arranging them according to their chemical properties and atomic weights. But his table had a number of prominent gaps. Mendeleev predicted that these represented unknown elements. He named them using the substance in the table sitting above the gap with the prefix eka, which is Sanskrit for the number 'one'. So, Mendeleev said, we should also have ekaboron, eka-aluminium, ekamanganese and ekasilicon.
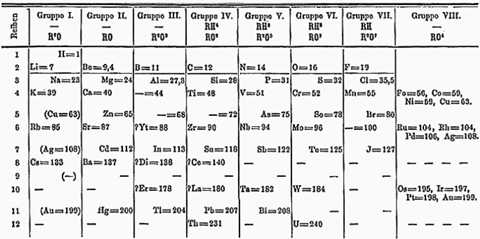
Of these, by far the most accurate prediction was for ekasilicon, occupying the slot we now give to germanium. Mendeleev came up with an atomic weight of 72, compared to an actual value of 72.6 from its four stable isotopes 70, 72 73 and 74. He was also pretty well spot on with its density and in predicting that it would have a high melting point - he even said it would be gray in colour.

It was seventeen years later, in 1886, that German chemist Clemens Winkler isolated the element from a newly discovered mineral called argyrodite, found in a mine near his home town of Freiburg in Saxony. Winkler first toyed with the name neptunium, after the recently discovered planet. But in 1877, a fellow chemist called Hermann had found a substance in the mineral tantalite which he believed was a new metallic element. Hermann had already taken the name neptunium for what later proved to be a mistaken finding. There was no new element in the tantalite.
Unaware of this mistake, Winkler decided to name his new element after his country. At the time, Germany was still relatively new, unified in the Franco-Prussian war in 1871. It might seem strange that he called his find germanium when Winkler knew his country as Deutschland, but the tradition was to use Latin names where possible, and the Romans had known much of the area as Germania, so this is where the element truly took its name from.
For a good fifty years, germanium was little more than a box on the periodic table. It really wasn't good for anything. It was only with the development of electronics that germanium's value as a very effective semiconductor came to light. A semiconductor is a material with conductivity between a conductor and an insulator, whose conductivity can be altered by an outside influence like an electric field or the impact of light.

The first use of germanium on a large scale was to replace the most basic electronic component, the diode. In the original valve or vacuum tube form, this had a heater that gave off electrons and an anode to which the electrons were attracted across a vacuum. It's like a one way flow valve in a water pipe - electrons can flow from the heater to the anode, but not the other way round.
As a semiconductor, germanium allowed the production of a solid state equivalent to the diode. Like most semiconductors, germanium can have impurities added to make it an electron donor – a so-called n-type material – or an electron acceptor, called p-type. By marrying p and n type strips of germanium, the element provided the same diode effect.

Germanium really took off with the development of the transistor, a solid state version of the triode valve. Here a small current can be used to control a larger one, amplifying a signal or acting as a switch. Germanium transistors were very common, but now have been replaced by silicon.
This is partly a matter of availability – as silicon in the primary constituent of sand, there's plenty out there, where germanium has to be mined at considerable expense. And silicon is a more effective semiconductor for making electronic components. But to have the effective silicon electronics we now depend on for everything from computers to mobile phones, requires extreme precision in purifying the element, which meant that silicon electronics weren't feasible on a large scale until the 1970s.
Once silicon took over, it might seem that germanium would be relegated to the backwaters of chemical obscurity as an also-ran that was no longer worth using. This has not happened because there are still applications where germanium is valuable, particularly in the specialist electronics of night vision equipment and as a component with silica in the fibre of the fibre optic cables used in communications.

Unlike many of the basic elements, there aren't many germanium compounds that have found a use. Germanium dioxide can be used as a catalyst in the production of the PET plastic used in many bottles, though it is rarely employed for this in Europe and the US. It is still primarily the pure element that has a role, if rather more specialized than it first was, in our electronics and communications. You may like to say it with flowers and give someone a gift of a geranium – but you're more likely to communicate down a modern fibre optic phone line, and then its germanium all the way.
Chris Smith
Brian Clegg with the story of germanium, which was named after the country it first came from. And speaking of elements named after countries, here's another one, although you'll have to look very hard to find it.
Peter Wothers
Whilst it is naturally occurring, or to be more precise, naturally formed – albeit briefly – during radioactive decay of other elements, the amount of francium on Earth is tiny. It has been estimated that at any one time there is less than a kilogram of the element in the entire Earth's crust.
Chris Smith
And bizarrely, despite being at the bottom of group one of the periodic table, francium isn't actually as reactive as caesium. And we'll hear why with Peter Wothers on next week's Chemistry in its element. I'm Chris Smith, thank you for listening and goodbye.













No comments yet