Meera Senthilingam
This week, beware the deadly odour, for it's a smell you may never forget. Wafting its chemistry into the air, it's Phillip Broadwith
Phillip Broadwith
What does death smell of? Crime novelists and war reporters alike refer to the 'smell of death' when encountering a dead or decomposing body. The characteristic odours of rotting flesh are generally recognised as being worth avoiding and difficult to forget once experienced. But what is it that makes a body smell so bad?
The chemistry of smell is incredibly complex. The human nose is able to distinguish many thousands of different molecules, sometimes at vanishingly small concentrations. But most of the smells we encounter are not caused by single compounds. The smell of a decomposing body is made up of all sorts of interesting compounds, but amines and sulfurous molecules make up the stinkier end of the spectrum. Most of those amines come from breakdown of the proteins in the corpse, and two of them have such fetid odours that they have been named putrescine - after the process of putrefaction - and cadaverine, after the Latin-derived word for a corpse: cadaver.
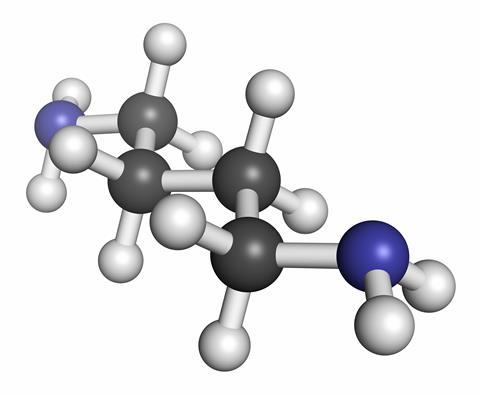
Putrescine and cadaverine are chemically very similar: they are both diamines. Both have short hydrocarbon chains with a primary amine group on either end. The difference is that putrescine has four carbon atoms in the chain between the two amines (its systematic name is butane-1,4-diamine), whereas there are five in cadaverine - or pentane-1,5-diamine.
This difference in structure derives from their biological sources - cadaverine comes from removing the carboxylic acid group from the amino acid lysine, whereas putrescine is derived from a different amino acid, arginine. The arginine is first transformed to ornithine, which is a key part of the urea cycle that allows the body to get rid of excess nitrogen. Some of that ornithine then ends up as putrescine and it is through routes like this that cadaverine and putrescine contribute, in small parts, to the smell of urine.
They also turn up in other bodily fluids - both contribute to the odour of semen, along with the related (and aptly named) polyamines spermine and spermidine, which are made by adding extra chains of three carbons, each ending with another amine group, to one or both ends of a putrescine molecule.
There will be amine-based smells anywhere where protein is decomposing,- that's why the poo of carnivorous animals like cats and dogs smells more pungent than that of herbivorous animals like rabbits or sheep: the protein content of their faeces is much higher.

But these diamines are not just about corpses and bodily smells. They are produced in normal living tissue as well; they seem to be used as cellular messengers or growth factors. Putrescine is also produced on industrial scale, although it is definitely not bound for the perfume industry! It is, in fact, made into plastics. As you may have heard in a previous podcast, the industrial process for making polyamide polymers like nylon is to take a diamine and another carbon chain with carboxylic acid groups at either end. Reacting these two molecules together makes long chain polymers, in which the diamine and diacid alternate, connected by amide bonds.
The original form of nylon, patented by the DuPont chemical company in the US, uses a diamine and a diacid that both contain chains of six carbon atoms. Hence it is called nylon 6-6. But changing the length of the linking carbon chain in either the diacid or the diamine can affect the properties of the final polymer, making it suitable for different applications. In 1990, for example, Dutch company DSM developed a polyamide called Stanyl, based on the same 6-carbon diacid but with putrescine acting as the diamine to make a polymer classed as nylon 4-6. The company claims that Stanyl is more resistant to heat and wear than nylon 6-6.
But where does DSM get its putrescine from? The image of the company harvesting smelly amines from a pile of rotting bodies in a basement somewhere belongs firmly in science fiction. DSM makes its putrescine from acrylonitrile and hydrogen cyanide. These two molecules combine together in the presence of a tertiary amine to make succinonitrile, which has the requisite four-carbon chain, but with the nitrogen atoms triply bonded to the terminal carbon atoms. Hydrogenation turns this colourless waxy solid into volatile, foul-smelling putrescine.
If I were running that reactor plant I'd want to be using up the putrescine as soon I could after making it - the smell of death is not one you want to linger.
Meera Senthilingam
Certainly not, as it may never leave. That was Chemistry World's Phillip Broadwith with the pungent, rotting chemistry of putrescine and cadaverine. Now, if this week's podcast has left an unpleasant sensation in your nose or mouth, fret not, because next week things get a lot sweeter - some might say too sweet.
Brian Clegg
It seems unlikely that the name of a chemical compound could also be an adjective. A few are twisted into adjectival form - watery, for example, sugary or salty. But the subject of this podcast is an adjective in its own right - and it's not a very positive one. Benzoic sulfilimine takes its common name from the word sacharrine (with an 'e' on the end), meaning 'excessive sweetness'. To call something saccharine is to suggest that it is too sweet; that it's sickly and unattractive. And yet this is exactly the property that made the world's first artificial sweetener such a success.
Meera Senthilingam
And to find out the chemistry causing saccharin to be so sickly-sweet, join Brian Clegg in next week's Chemistry in its element. Until then, thank you for listening, I'm Meera Senthilingam.













No comments yet