A form of microscopy is shaking up nanoscience research and forcing scientists to reconsider many established theories. Emma Davies investigates cryoTEM
A form of microscopy is shaking up nanoscience research and forcing scientists to reconsider many established theories. Emma Davies investigates cryoTEM
Cryogenic-temperature transmission electron microscopy (cryoTEM) has long been used by biologists but colloid and material scientists are now starting to take advantage of the extra dimension that it gives their research.
Ishi Talmon, a researcher in the department of chemical engineering at the Technion-Israel Institute of Technology, Israel, pioneered the technique for non-biological systems. He started using cryoTEM in 1976 when studying for his PhD on microemulsions at the University of Minnesota, US, and is still one of the lead researchers in the field.
It took a long time before other researchers in non-biological fields latched on to cryoTEM’s attractions. Those who use it are frequently astounded by the results. Darrin Pochan, a materials researcher at the University of Delaware, US, started working with the technique in 1999 and has found it gives completely unexpected results, even in established areas such as lipids and surfactants. ’People think that they know what is going to happen. They throw samples in a cryoTEM and take some images and all of a sudden they see structures that are completely unpredicted.’
TEM takes off
Last year Pochan did a quick search of cryoTEM papers for a talk at a meeting organised by the Materials Research Society, US. He discovered that in 2000 only a handful of articles were published on the technique but by 2004 the number had grown by at least an order of magnitude. He assumes that the number will continue to grow as more researchers take up the technique. ’People already have electron microscopes. You just need the additional peripheral stuff,’ he says.
Talmon agrees that cryoTEM is becoming more popular and attributes this to ’the advance of bio- and nanotechnologies and especially the combination of the two’. He echoes the words of many colloid scientists who had been working on nano systems for many years before the term nanotechnology was coined. ’We have been doing nano research for 25-30 years but we didn’t know it was nano until recently,’ he says. ’Suddenly it occurred to us that we had been doing nano all the time and in a way we had a technique that suddenly had so many more applications, with so many more potential users.’
CryoTEM explained
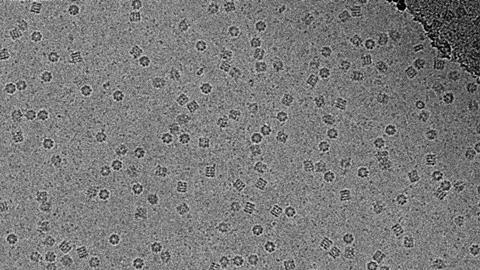
Transmission electron microscopes (TEM) use electrons instead of light. A source at the top of the microscope emits electrons that travel through vacuum. Electromagnetic lenses focus the electrons into a very thin beam, which then travels through the sample. Electrons left unscattered by the sample hit a fluorescent screen to give a shadow image.
In cryogenic-temperature transmission electron microscopy (cryoTEM), thin films of the sample liquid are cooled at ultra-fast rates before being transferred to the transmission electron microscope and maintained at cryogenic temperature. A small drop of the sample is applied to a TEM grid held inside a controlled environment vitrification system (CEVS). Blotting with filter paper gives thin liquid films that span the holes in the grid.
The films should be no thicker than about 300nm and of very low vapour pressure to make them compatible with the high vacuum conditions and the limited penetration power of the electron beam.The temperature and atmospheric composition in the CEVS are controlled to minimise volatile loss and to allow quenching of the liquid.
The grid is then plunged into a cryogen such as liquid ethane at -170°C. This vitrifies the sample with a cooling rate of about 10,000°C per second. It is then stored under liquid nitrogen and transferred to a TEM cooling holder. The sample must be kept at cryogenic temperatures to avoid phase change and sublimation of volatiles.
Small beginnings
Talmon admits that he launched into using cryoTEM in a naïve way. ’Soon enough we understood that we didn’t have the right equipment or the right approach and we knew nothing about the interaction between an electron beam and a vitrified specimen,’ he recalls.
He set himself a challenge to come up with a working technique. He didn’t succeed in studying microemulsions using cryoTEM. ’But we learned a lot and we got our very first reliable images for some structures in ice.’
Several years later his group started using a technique developed by Jacques Dubochet, a biologist who currently works at the University of Lausanne, Switzerland. ’He was the first person who showed how to flash-freeze or vitrify thin layers of water and that was a major breakthrough,’ recalls Talmon. Dubochet’s technique, which preserves sample structure, gave Talmon the results that he wanted and the team was finally able to visualise surfactant structures such as micelles and vesicles. One of his greatest achievements was to obtain direct images of threadlike micelles, which were later identified as intermediates in the solubilisation of lipids by surfactants and the reconstitution of micelles by dilution. The findings did not agree with suggested theories at the time and were at first rejected by the biophysical community. ’The problem is that people have their pet theory and model and when confronted with the real image, they find it difficult to accept,’ says Talmon. Fortunately, his findings were backed up by small angle neutron scattering experiments.
Micellar magic
Pochan works mainly on molecular self-assembly in solution. Until he started using cryoTEM he was restricted to analysing his systems using indirect methods such as light scattering or neutron scattering. ’In the past you would have your straightforward goal of a structure you’d want to see. You would do your scattering experiment and say OK here’s what we think the structure is. But now, even within one system, you’ve got your structures you can make but then if you slightly tweak the conditions you can get those structures to start to change to other things,’ says Pochan.
Some interesting structures have emerged from Pochan’s work with amphiphilic polymers which self-assemble in solution to form micelles. ’One thing we’re starting to see is that when the micelles transition from one shape to another, depending on a solution stimulus, they go through a lot of intermediate stages that you’d never be able to see with a scattering technique, for example, and you certainly wouldn’t be able to predict because they’re very, very strange transitions.’ These intermediate structures have unusual appearances. For example, Pochan describes some of them as ’very big pancakes with hairs coming out of the ends’. Others look like mushrooms growing on top of one another, he says.

Pochan has also worked on systems that form stable disc-like micelles that had been predicted but rarely seen. Now the researchers have set up a very robust system that makes discs reproducibly, says Pochan. They have also made toroidal ring-like micelles and used cryoTEM to confirm that the structures are stable.
Pochan’s team is now using cryoTEM to study gel networks. The properties depend on the local nanostructure of the self-assembled molecules. He uses cryoTEM to characterise peptide hydrogels. ’This is proving challenging but we’re slowly starting to be successful,’ he says. The main problem has been preparing gel samples thin enough for electron beams to pass through. Vitrifying the gels makes them very brittle and liable to fracture. So the researchers are trying several different ways of applying very thin films of the gels to TEM grids.
Frank Bates, head of chemical engineering and materials science at the University of Minnesota, US, also uses cryoTEM to study polymer systems. He started using the technology in 1997 and says that his group has obtained ’many outstanding results with the technique and sometimes very surprising ones’.
One of the surprising discoveries came from work on aqueous dispersions of block copolymer surfactants. The researchers knew that relatively low molecular weight diblocks produced spherical and cylindrical micelles and vesicles. When they increased the overall diblock copolymer molecular weight, Bates expected to see larger micelles with the same geometric shapes as the smaller versions. What he discovered was far more exciting: a new structural element called three-fold branching. Bates says that this discovery is important because it demonstrates that an increase in molecular size causes a qualitative structural change. ’Branches lead to network formation at higher concentration,’ he says.
The results were widely accepted by the scientific community and have stirred healthy debate regarding the self-assembly of block copolymers in water, notes Bates. He considers that this is because, ’unlike indirect characterisation techniques such as spectroscopy, all forms of real-space imaging including transmission electron microscopy generally are easily interpreted and often are less subject to interpretation by specialists’.
CryoTEM is also helping studies of the most famous nanostructure, C60, and a group from Rice University, Texas, US, has used it to observe C60 aggregates in water as a suspension. According to John Fortner, a member of the research team, they had some concerns that the crystalline nature and dimensions of the aggregates observed via traditional dried TEM were in fact an artefact of the drying process. ’The cryoTEM results verified that the materials in suspension were exactly as they appear via traditional TEM,’ says Fortner. ’It’s a nice technique if you can get it to work,’ he adds.
Hans von Berlepsch, from the Free University Berlin, Germany, is another keen cryoTEM user. His research team has used the technique to study a special class of self-assembled structures called J-aggregates, which form spontaneously from certain cyanine dyes in aqueous solutions. He considers that cryoTEM is the best way to observe these structures without distortion.
Most studies had ascribed the optical properties of J-aggregates to a brickwork arrangement of the chromophores. ’When we entered the field by studying a new class of J-aggregates we faced big problems to explain our light scattering results,’ he told Chemistry World. CryoTEM answered their questions: ’We were highly surprised to detect several different structures such as tubular aggregates, ribbons, superhelical bundles and other unexpected morphologies’. The researchers now have a deeper understanding of the complex excitonic absorption spectra of tubular J-aggregates and have attributed the chirality of some of the structures to ’helical packaging architecture’.
Von Berlepsch’s team has also used cryoTEM to study fullerene and calixarene derivatives and has been able to characterise the micelles with ’unprecedented precision’. These micelles form nanocontainers which can be turned on and off by an external stimulus.
Companies
FEI, Oregon, US, sells a range of transmission electron microscopes (TEM). In 1997 it merged with Philips Electron Optics, a Philips Electronics unit.
JEOL, originally set up as the Japan Electron Optics Laboratory, is based in Tokyo. The company sells a range of TEM systems.
Leica Microsystems, part of Leica, is based in Wetzlar, Germany. The company sells electron microscopy accessories such as cryochambers, imaging systems and software.
Gatan is based in California, US. The company sells TEM software and laboratory equipment including digital charge coupled device cameras, sample preparation tools and cryo holders.
The Nano technology systems division of Carl Zeiss is based in Germany and sells a range of energy filtering TEMs and accessories.
Oily colloids
Much of the cryoTEM work so far has been on water-based systems but Talmon has also studied non-aqueous samples. He has found himself limited to certain solvents because many, such as linear paraffins, cannot vitrify. Most apolar solvents such as hydrocarbons dissolve in the standard cryogen, liquid ethane.
In some systems such as those with a siloxane solvent, electron beam damage is so severe that even using low-dose techniques is unsuccessful. Talmon’s team has succeeded in imaging unusual non-aqueous systems such as lecithin dispersions in soya bean oil. The researchers have discovered that the triglycerides found in many natural oils act as ’cryoprotectants’ that allow oil systems to vitrify in liquid nitrogen.
Drawbacks
How long does it take to get the cryoTEM working for a new system? Pochan laughs: ’It takes a lot of practice. It is a very technologically dependent technique and I guess what I mean by that is if you don’t have all the right pieces like a good low accelerating voltage microscope with a sensitive CCD [charge coupled device] detector, a nice holder with a nice transfer stage and a nice piece of equipment to vitrify liquids and gels to image, then it becomes extremely difficult.’ Bates also considers that ’cryoTEM can be quite challenging’, but says that ’once the various experimental methods are mastered this technique offers a unique glimpse into the structure of many forms of soft materials in water’.
Bates considers that the primary concern with cryoTEM is the presence of artefacts that can arise during sample preparation. He lists a few reasons why this might happen: ’Firstly, the structures examined are confined to a thin film, usually 200 to 300nm in thickness. Therefore, 3D features may be distorted. Also, the film may not be uniform in thickness. Secondly, in forming the thin film, excess water is blotted from the microscope grid and this can impose shear forces that distort the structures. Thirdly, the water may crystallise in places thereby disrupting the material. And finally the electron beam may deposit excess energy in the specimen leading to localised damage.’

Positive future
Talmon considers that cryoTEM has a good success rate. ’It doesn’t take that much to train say a student or a postdoc. Within six to eight months, I would say, we can take a good graduate student and get him working independently on a cryo system.’ But, he cautions, it takes the supporting environment of professors, postdocs, and full-time researchers who work with them to allow those people to pick up the technique in such a short time.
Bates thinks that the outlook for cryoTEM is good. ’Through much experience, particularly seminal contributions by others including Ishi Talmon, I believe the scientific community has very much accepted the viability of the method,’ he says.
A few years ago Talmon collaborated with the late Nobel prize winner Richard Smalley from Rice University, US, to image nanotubes suspended in micellar solution for him. At a seminar when the results were presented, Smalley commented that the technique ’opens the eyes of those who use it’. Given its rapid growth, it is bound to open many more eyes.
Further Reading
-
V Moore et al, Nano Lett., 2003, 3, 1379
-
J Fortner et al, Environ. Sci. Technol., 2005, 39, 4307
-
M Adrian et al, Nature, 1984, 308, 32
-
E Kesselman et al, Macromolecules, 2005, 38, 6779
-
D Danino et al, J. Colloid Interface Sci., 2002, 249, 180
-
S Jain and F Bates, Science, 2003, 300, 460
-
D Pochan et al, Science, 2004, 306, 94
-
H von Berlepsch et al, J. Phys Chem B, 2004, 108, 18725












No comments yet