The increasing sophistication of detection techniques means mass spectrometry can now escape the laboratory. Emma Davies sets sail to new horizons
The increasing sophistication of detection techniques means mass spectrometry can now escape the laboratory. Emma Davies sets sail to new horizons

The ‘intelligent knife’ could help the surgeon to avoid cutting directly into tumours, reducing the risk of leaving behind bits that could spread the cancer to other parts of the body. It could also warn of the presence of tumour cells at the edge of the section of tissue being removed.
Analytical chemistry has already proved useful in tissue characterisation. For example, matrix-assisted laser desorption/ionisation (Maldi)–mass spectrometry and nuclear magnetic resonance spectroscopy can provide valuable information on tissue type. Yet neither technique can give unambiguous tissue identification in real-time.
In 2008, Takáts had a brainwave while working at the University of Giessen in Germany. He realised that – just like Maldi – the electrosurgical tools found in just about any operating theatre in the world ionise molecules. Whereas Maldi uses a UV laser to produce a hot plume of analyte molecules which are then protonated, the surgical tools used for dissection or to stop bleeding use heat generated by an electric current. ‘They thermally disintegrate tissue and evaporate tissue material,’ says Takáts.
Combining the electrosurgical tool with MS gives the ‘ultimate tissue detection device’, he says. Now known as rapid evaporative ionisation mass spectrometry (REIMS), the electrosurgical–MS combination gives metabolic fingerprints of tissues. ‘There is plenty of evidence from Maldi imaging and other imaging methods that metabolic fingerprints can be used to identify particular types of tissue. One can recognise not only cancer but sub-types of different cancers,’ says Takáts. The tissue analysis is based on differences in phospholipid distribution.
Takáts does not adapt the electrosurgical tools but has spent over three years ‘heavily modifying’ the MS. ‘The first challenge was how to get the ionic species from the surgical site to the MS – a distance of at least two metres,’ he says. The researchers opted for a Venturi air pump and fluorinated polymer tubing, which can transfer ionic species to the MS over a distance of up to six metres.The data interpretation, which takes a few milliseconds, gives audiovisual feedback to the surgeon
A second challenge was to make the MS compatible with the sterile surgical environment. The team built a chassis insulated for noise and electromagnetic radiation. They also had to keep large non-charged material out of the instrument. ‘There are big droplets and pieces of tar flying through the tubing that we don’t want to be introduced into the MS,’ says Takáts. ‘We use some momentum separation and then get rid of large particles and keep the instrument as clean as possible.’ All of the air leaving the MS is filtered to remove any infectious agents and dust.
Then there is the significant matter of sorting rapidly through vast amounts of data to give metabolic fingerprints. Takáts’ team uses a statistical technique called principal component analysis followed by linear discriminant analysis. The researchers have built up a database of almost 100,000 chemical entries. Depending on the surgery type they can select the relevant part of their data, such as that relating to the liver.
Takáts only recently moved his research group to Imperial’s Department of Surgery and Cancer. He has already successfully tested his new technique in vivo in his native Hungary and in Germany; the results are in press. He now has a new instrument installed in a room adjacent to the operating theatre at St Mary’s.
The new tests at St Mary’s show that the instrument can be used continously for four hours, a significant improvement on early versions of REIMS, which only worked for a couple of minutes before requiring cleaning. The current set-up can give a metabolic fingerprint in 0.7 seconds, says Takáts. ‘Overall the ion transfer mass analysis takes something like 0.2 to 0.5 seconds. The data interpretation, which takes a few milliseconds, gives audiovisual feedback to the surgeon.’
Takes your breath away
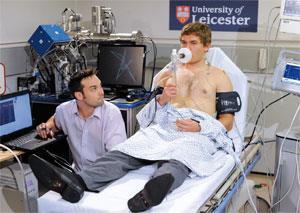
The DDU uses a range of analytical techniques, from thermal imaging systems to blood flow and oxygenation monitors. The idea is that patients entering A&E will be screened to give quicker and more patient-specific diagnoses.
‘Breath is actually quite a challenging analyte because it is very humid and transient,’ says Monks. ‘You have to get the analytical technique to be insensitive to very high humidity and you have to make sure that the water doesn’t condense out because many of the volatiles that you might be interested in are water-soluble.’
Monks’ team analyses breath in real time using proton-transfer reaction (PTR) time-of-flight MS. Protonated water mixes with sample molecules inside a drift tube and transfers protons to those volatile organic carbon compounds (VOCs) with a greater proton affinity than water. ‘The interesting thing about that is that it’s a chemically controlled reaction so it’s very quantitative. We can actually work out the concentration [of a VOC] on somebody’s breath,’ says Monks.
‘With time-of-flight MS we get all of the masses, all of the time. And that’s important because we are in discovery mode with DDU.’ Monks’ team uses statistical analysis for pattern recognition to create chemical fingerprints.
Another challenge is capturing the breath in a regularised manner. The first part of the breath, which is from the nose and mouth, is of no interest. ‘We want breath from the lungs because that’s where the VOCs will have exchanged from the blood,’ says Monks.
VOCs are analysed over a series of ‘very reproducible’ breaths collected using what Monks calls a magic wand – a tube with a mouthpiece. The wand measures levels of carbon dioxide to give a marker so that researchers can be sure that they are always sampling the same amount of breath at the same part of the breath cycle. The breath is then transferred two metres to the MS in heated tubing to prevent condensation.
Monks’ students are currently building up a chemical database to build a baseline chemical picture for different types of people. The idea is to estimate ‘normal’ levels of key VOCs for different types of people in different age bands, so they can spot anything out of the ordinary. By the end of 2012, his team will have collected data on 500 people in A&E.We can actually work out the concentration of a volatile organic carbon compound on somebody’s breath
‘We need to find out what the biomarkers are for the disease state. Once you have that, you can build a cut-down device, maybe hand-held even, that you can then use in a hospital environment for a single diagnostic test,’ says Monks. ‘That would be a long-term goal.’
Early results are promising. In a small study of children with cystic fibrosis, Monks’ team has been able to spot those with active chest infections using MS based on breath analysis. The VOCs given out by microbes in the lungs are different to those free from infection, explains Monks.
Monks has also done several in vitro studies using PTR-MS to analyse VOCs given off by the drug-resistant bacteria MRSA (methicillin-resistant Staphylococcus aureus). ‘We can actually tell which are the active strains of MRSA and which are the non-active strains from VOCs. The ones that are clinically active have a different VOC signature to the ones that aren’t,’ adding that his team has compared some fungal and bacterial VOCs, and picked up differences in signatures.
A hostile atmosphere

Hackenberg collects and analyses air samples at regular intervals, looking in particular for terpene emissions. The air is then scrubbed to remove all the VOCs before passing it through the sea-water to collect the volatiles. Andrews and Hackenberg only had four days to set up the GC–MS and to make it seaworthy. ‘You don’t know what space you’re working in until you get there and we are working in a wet lab where people are throwing water around,’ laughs Andrews. ‘Vibration, seawater, and humidity: the worst environment for a GC–MS.’ The machine ‘floats’ on shock mounts inside a flight case which is strapped firmly to the ship.
Andrews has also been involved in aircraft projects led by Alastair Lewis, also of the University of York, monitoring VOCs given off by forest fires in Canada. A GC–MS is installed in the aircraft for air analysis in almost real time. Taking the GC–MS on a flight presents extra challenges, including the difficulty bringing in air samples from outside the aircraft and compensating for changes in pressure with altitude. But overcoming these problems is worth the effort to be able to detect complete ‘unknowns’ and identify new species.

‘The hardest thing for us is the environments that we work in, because the equipment is designed to work in the lab and none have the performance that we require,’ says Andrews. Expeditions are painstakingly planned for many months. While on an expedition, an atmospheric researcher is often fully responsible for their own bit of kit and will be expected to take along spare parts. When the kit breaks, they need to know how to take it apart and fix it.
Do it yourself

All of Barran’s MS systems are home-built. ‘It’s really common in my community to make adjustments to instruments to do things that are not commercially available,’ she says. Barran did her PhD with Tony Stace and Nobel prize-winner Harry Kroto, who made key discoveries using homebuilt mass spectrometers. Barran soon learnt how to take apart and build an MS and surprised herself with how much she enjoyed the task and how good she was at it.
‘When I teach undergraduates mass spectrometry I have a lecture called “Beneath the white boxes”,’ she says. ‘I think there is increasingly this white box attitude.’ The trouble with MS, she adds, is that it’s a destructive and dirty technique and the boxes need to be opened and their contents taken apart and cleaned.
Barran has applied and been turned down for funding for a new commercial machine, largely because of the eye-watering costs involved. Restrictions on resubmitting the same funding proposal make life difficult. ‘If you’re an instrument development person, it’s very difficult to come up with a wholly new idea,’ she says.
Barran is hoping for a resurgence in device development in universities, driven by advances in electronics. Her team is now able to build an ion mobility mass spectrometer with a higher resolution than provided by leading manufacturers – for about £20,000.
Those wedded to MS as an analytical tool commonly show high levels of creativity and technical ability, taking the technique to an ever-expanding list of applications, from airports to hospitals. Before long, one can imagine home MS kits for various types of chemical detection. A nice idea – provided they come with a manual.
Emma Davies is a science writer based in Bishop’s Stortford, UK
Further information
The University of Leicester’s Diagnostics Development Unit
Atlantic Meridional Transport (AMT) programme
Z Takats et al, Future Oncology, 2012, 8, 113 (DOI: 10.2217/fon.11.151)
P E Barran et al, Anal. Chem., 2008, 80, 6336 (DOI: 10.1021/ac800651b)












No comments yet