Now a biological reality, researchers are starting to figure out the many roles of left-handed nucleic acids such as Z-DNA – from immunity to controlling our genetics. Rachel Brazil reports
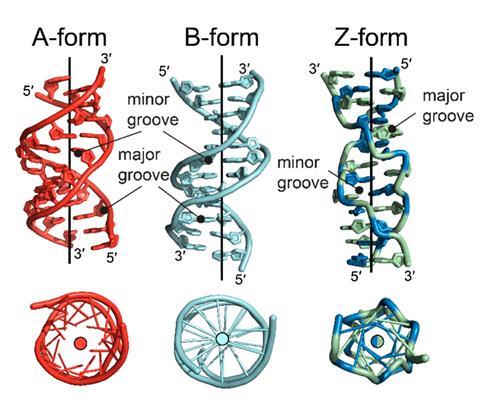
Biology encodes its genetic instructions in sequences of base pairs within the backbone of DNA’s right-handed double helix and through the RNA molecules subsequently transcribed. But what if the helices of these nucleic acids twisted in the opposite left-handed direction? It’s a mistake sometimes spotted in stock illustrations of the famous molecule, but a crystal structure for the left-handed DNA conformer, known as Z-DNA, was published in the 1970s. It looks a little different to the right handed version – rather than a smooth corkscrew, the backbone forms a zig-zag shape, giving rise to its name. Investigations led to suggestions that Z-nucleic acids could play a role in biology, but these were widely met with scepticism.
In the last five years, clear evidence has shown that both DNA and RNA do form left-handed double helices in biology, and that these structures play a role in our immune responses to viral infection and inflammation. Z-DNA may even have a fundamental role in genetic control. Now a growing community of researchers are looking at ways this new biology can be exploited to fight cancer and other diseases.
First glimpse
The Z-nucleic acid story starts in the MIT lab of biophysicist Alexander Rich (now deceased), best known for solving the structure of the transfer RNA molecules that carry amino acids in protein synthesis. By the 1980s his lab had discovered that regular right-handed B-DNA could flip into the usually less thermodynamically favoured left-handed Z conformer, particularly at high salt concentrations. Changing the direction of the helix does not produce a mirror image of B-DNA, because in the left-handed version the bases alternate between the anti position, where the base points away from the sugar ring, and the syn position, where the base projects over the sugar ring, rather than only the anti position as in B-DNA. This leads to a structure with even grooves between the two helices and a zig-zag appearance. Rich’s group also discovered that where the direction of its spiral changes, at least one unpaired base is extruded outside the helix, known as a B-Z junction.
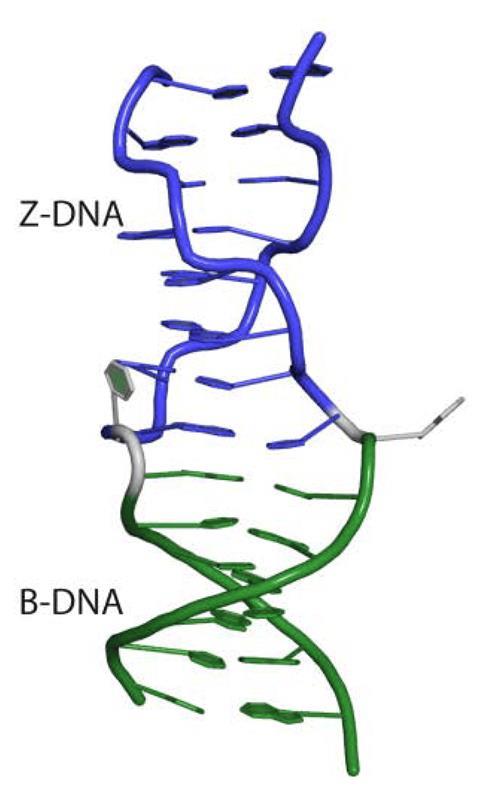
But proving Z-nucleic acids exist in nature and have a biological function was more of a challenge. Using an antibody probe that recognised Z-DNA, they found stretches in plasmids (small circular DNA molecules) that could flip into the Z conformation in sections where the base-pairs have repeated alternating purines and pyrimidines motifs, particularly the purine guanine (G) and the pyrimidine cytosine (C).
Hundreds of such sequences of up to 100 base-pairs were identified in the human genome, often clustering around promoter regions where gene transcription is initiated. Researchers showed that Z-DNA formation is more thermodynamically favoured during active transcription, behind the opened up DNA where there are regions of under-coiled double stranded DNA (known as negative supercoiling). Another piece of the puzzle was the discovery of several proteins containing a domain, named Zɑ, that binds to the double stranded Z-nucleic acid backbone, both to Z-DNA and double stranded RNA which can pair with itself in highly repeating sequences and take on the Z-conformation.
Rich and his colleagues suggested Z-DNA acted as a transcription control, with sections of DNA flipping between the two conformers, dynamically switching genes on and off. ‘Everyone was excited about it,’ says immunogeneticist Alan Herbert, who at that time was a researcher in Rich’s lab. But he says, ‘the data didn’t support this really simplistic model’. There was a long-running debate, which Herbert says was not helped by the academic politics surrounding the RNA field which left Rich lacking allies. By 2000 the idea had lost momentum, with many seeing Z-nucleic acids as just a curious chemical anomaly with little relevance to biology. ‘I would go to meetings and put posters up on the Z-DNA binding protein I found and the world would walk by, with eyes averted, looking the other way,’ Herbert remembers.
This all changed in 2019, when Herbert, now the founder of InsideOutBio, a DNA therapeutics company based in Massachusetts, US, had some time to return to the area and found that he could directly link mutations in one of the Z-DNA-recognising proteins he discovered to several serious neurodevelopmental conditions. Aicardi–Goutieres syndrome and bilateral striatal necrosis/dystonia are both linked to an inability to regulate immune signalling molecules called interferons. Losing the ability to bind Z nucleic acids could lead to serious consequences, showing once and for all that Z-nucleic acids were present in biology.
Innate immunity
‘One thing that’s really been quite exciting over the last five years is the realisation that this unusual Z-confirmation triggers innate immune responses,’ explains Jan Rehwinkel, an immunologist at Oxford University in the UK. These are the fast first line defences our bodies raise against infection or other types of cell dis-regulation. One way it does this is through nucleic acid sensor proteins that can detect viral nucleic acids in all cells and set off signalling pathways via the molecule interferon 1, eventually leading to the death of infected or damaged cells.
It turns out that a lot of circular viral nucleic acids have stretches that are prone to flip to the Z conformation and the immune system has adapted to sense this with a protein called Z-DNA binding protein 1 (ZBP1), which contains two Zɑ domains, each about 80 amino acids, that adopt a winged helix-motif that specifically bind around any Z-nucleic acid backbone and trigger an immune response. Rehwinkel proved this using mice engineered with mutated ZBP1 proteins that were unable to bind Z-nucleic acids. On viral infection, their signalling and cell death pathway did not function.
But on top of viral infection, Z-RNA is also produced by cells themselves when infected or under stress due to inflammation. ‘There’s lots of what’s called endogenous retro elements in mammalian genomes, including the human genome,’ say Rehwinkel. ‘These are [short DNA] remnants from retroviral infection from millions of years ago, and many of these are repetitive, and there are plenty of them.’ Millions of copies of these highly repeating sequences are dispersed throughout the human genome.
When the cell gets stressed, it starts inducing expression of these regions which form Z-RNA. ‘Viruses often disrupt the termination of the RNA message and so suddenly, you’re transcribing regions that are not normally transcribed in the cell,’ explains Herbert. Other diseases can also cause high levels of Z-RNA, which then leads to more production of the Z-sensor protein ZBP1 and a greater immune response. Rehwinkel thinks this Z-RNA plays an important role in an array of health conditions including autoimmune disease and cancer.
But human biology has another trick up its sleeve to prevent this. The other human protein discovered so far that recognises Z-nucleic acids is an RNA editing enzyme, ADAR (adenosine deaminase acting on RNA). It edits the base adenosine (A) in double-stranded RNAs to inosine, which is chemically similar to guanosine (G) and so disrupts the RNA’s base pairing. By doing this it stops the RNA binding to itself, removing the double stranded Z-RNA and the immune response it causes. In 2022 a series of publications using different methodologies confirmed this mechanism.
Fighting cancer
Philip West, an immunologist at The Jackson Laboratory for Mammalian Genetics in Maine in the US, has shown another way Z-nucleic acid sensing occurs in cells. His work focuses on what happens when mitochondrial DNA leaks into a cells’ cytoplasm from the mitochondria, which is another sign of cell damage. The presence of mitochondrial DNA in cytoplasm has been linked to heart damage caused by the common chemotherapy drug doxorubicin. When this happens it can disrupt chemotherapy but it also has long term consequences, explains West. ‘A large number of people on therapy experience no effect and then sometimes 30 years later develop problems with their heart function and can go on to have heart failure.’
The exciting thing for us is that we’ve identified Z-binding protein zbp1 as a player
‘It turns out that mitochondrial DNA looks very similar in many regards to bacterial or viral DNA, given that it’s a circular genome,’ says West. ‘[So] we thought that this ZBP1–interferon pathway could be playing a role in chemotherapy-induced cardiotoxicity.’ Previous work had also shown that doxorubicin, which can intercalate between DNA bases, causes the kind of supercoiling that leads to DNA flipping to the Z form and West showed there are increased levels of Z-DNA in doxorubicin-treated cells.
He then used genetically engineered mice lacking ZBP1 and the interferon 1 signalling molecule and found they were protected from doxorubicin-induced cardio damage. ‘The exciting thing for us is that we’ve identified [Z-binding protein] zbp1 as a player,’ says West. His long-term goal now is to see if a therapy can be designed to reduce the impact of mitochondrial Z-DNA in heart cells, without switching off the immune pathway in tumours where it might have a positive contribution to destroying cancer cells.
Herbert is now focusing on how Z-nucleic acids could be harnessed to fight cancer. Misregulated gene expression in cancer cells also leads to a build-up of Z-RNA. Normally this would be sensed by ZBP1 which would lead to cell death, but many tumours have evolved mutations that allow them to harness the production of the ADAR enzyme to protect themselves, by editing away Z-nucleic acids and in doing so, suppressing any immune response. In cell culture, over 40% of cancers have been found to be dependent on ADAR for survival.
The solution devised by Herbert was to look for a small molecule that might be able to trigger regions of DNA to flip to the Z configuration and produce more Z-RNA, so overriding the ADAR editing process, and activating an immune response. As healthy cells produce minimal amounts of the Z-nucleic acid sensing protein they will be less vulnerable to this cell death pathway. ‘When you have tumour cells that have this inflammatory environment going on, these guys are really vulnerable, so if you can activate a cell death pathway in tumours, then you have quite a good therapeutic window,’ says Herbert.
Collaborators working with Herbert, led by Sid Balachandran at Fox Chase Cancer Center in Philadelphia in the US, identified a Curaxin, a tricyclic carbazole drug candidate already noted to have anti-cancer properties. So far they have tested the approach with a subset of melanoma cells called cancer-associated fibroblasts (CAFs) which are known to express the Z-DNA binding protein. These cells are usually resistant to the immunotherapy drugs that help kick-start the patient’s own immune system, but when given alongside Herbert’s new Z-inducing molecule, the brake on the immune system is lifted, leading to cancer cell death.
Targeting the pathways that regulate Z-nucleic acids is an entirely new way to treat cancer. ‘That is a really exciting development,’ says Rehwinkel,
Genetic instability
Cancer researcher Karen Vasquez from the University of Texas at Austin has come to study Z-nucleic acids from a different perspective. Her lab is focused on understanding the genetic instabilities that can lead to cancer in the first place. She found that sections of Z-DNA are recognised by the ‘mismatch repair’ mechanisms that usually corrects post-replication base pair mismatches. In this case it recognises the extruded bases that make the BZ junction between the B-DNA and flipped Z section as a mismatched loop in need of repair. The cell’s repair machinery then tries to correct the mistake eventually leading to a double strand break.
Why would Z-DNA be there if it’s just causing problems?
‘We looked at 20,000 different human cancer genomes and found using bioinformatics and algorithm design that Z-DNA is highly enriched at mutation hotspots in human cancer genomes,’ says Vasquez. The breakages are leading to genetic instability through deletions and translocations – where faulty repair leads to DNA from two different chromosomes joining up. These types of errors can ultimately lead to cancer.
One question Vasquez asked herself is why do these cancer-causing Z-DNA elements remain in the genome – in abundance – rather than being selected out? ‘Why would they be there if they’re just causing problems?’ asks Vasquez. The answer may lie in their ability to facilitate evolution. Vasquez began to see where the benefits might be after she was approached by Stanford developmental biologist David Kingsley to explain his recent results involving the evolution of stickleback fish as they move from freshwater to saltwater.
These fish have changed environment many times over the last 15,000 years and tend to lose their pelvic hind fins when this happens. Previous work has identified the pelvic enhancer gene involved in the changes and Kingsley wondered if these rapid evolutions were linked to double strand breaks caused by Z-DNA. ‘We found indeed, that sequence did form Z-DNA, it did induce deletions, and it did cause the gene alteration that led to that fin development.’ Vasquez says the double strand breaks leading to large deletions are likely to explain the enhanced mutation rates that allow the fish to repeatedly adapt and likely shows why Z-DNA can have a long-term positive impact.
Back to transcription?
After 40 years, Herbert is now thinking back to Rich’s original ideas linking Z-DNA to transcription. He has named the regions of the genome that form Z-DNA ‘flipons’, and his most recent collaboration uses an AI model to predict where they occur. They uncovered 290,071 segments making up 3,167,809 base pairs (0.16% of the genome). Herbert now thinks the flipons occurring near gene-promoter regions act as actuators. When they flip from B to Z, they capture the chemical and potential energy released when RNA polymerases prise open double stranded DNA during transcription. They can then release this energy when they flip back, which Herbert suggests could be used to power the assembly of other complexes involved in the packaging of DNA, or to reset promoter regions by removing any bound proteins and allow transcription to be re-initiated. There is still a lot to work out and evidence to find, but ultimately he thinks this will lead to a new understanding of our genetic machinery and new therapeutic strategies for a wide variety of diseases.
Another still unanswered question for those working on Z-RNA is whether it forms independently or only when stabilised by Z-binding proteins. The ‘chicken and egg’ question is being investigated by University of Houston RNA expert Quentin Vicens and NMR chemist Beat Vogeli from the University of Colorado. Vicens thinks that the Z conformer might be more common in RNA than we think. ‘We have enough evidence that there are plenty of RNAs in our cells that adopt these stretches of Z,’ says Vicens. The NMR work with Vogeli shows a broad range of RNA sequences, even those devoid of GC repeats.
A few years back it wasn’t quite so clear whether Z-nucleic acids have any role in biology
They are building up a picture of a dynamic process with RNA flipping into the Z conformation for short periods of time. Using NMR, Vogeli can detect minor states and can clearly identify Z-RNA with its distinct spectrum. ‘What we can say right now is that if there is a Z form present in normal conditions, then it will be less than 1% of the time,’ says Vogeli.
The team are also hoping to image how the Z alpha domain binds to Z-RNA. ‘No one has [yet] seen a structure of a complex between one of these Z binding proteins bound to some biologically relevant RNA,’ says Vicens.
Vasquez and others are also keen to detect Z-nucleic acids dynamically in cells. ‘We’re working on ways to do this, with fluorescence-based assays to look at [it occurring naturally] in a cell,’ she explains. Using fluorescence resonance energy transfer (FRET) technology she can monitor Z-DNA formation using a fluorophore and quencher attached to positions on the DNA molecules that only move close enough to quench the florescence when Z-DNA forms. ‘[We] can do it on plasmids right now,’ says Vasquez, but her team is still working out how it might be done on genomic DNA and hope to use the method fora high-throughput screen of small molecules that might be able to flip sequences from B to Z or vice versa.
After many years of neglect, new insights into Z-nucleic acids are now coming fast, but it’s not the only non-B-DNA nucleic acid structure to be getting attention. Research is showing that G-quadruplexes – four-stranded DNA secondary structures – are implicated in transcriptional regulation and cancer, and triple-stranded DNA (also known as H-DNA or triplex-DNA) has also been linked to gene modulation. ‘A few years back it wasn’t quite so clear whether Z-nucleic acids have any role in biology, but [now] that’s become quite clear, it will be exciting to see whether the same is true for G quadruplex, or other unusual three-dimensional structures that nucleic acids might adopt,’ says Rehwinkel.
Rachel Brazil is a science writer based in London, UK
















No comments yet