Thousands of papers have been published on curcumin’s healing potential, but its usefulness is not yet proven, finds Andy Extance
Every year 750 in every million children are born with extremely distressing and potentially blinding clefts in an eye, in an ailment known as ocular coloboma. Yet curcumin, the major component of the spice turmeric, has been shown to stop the cell death that causes it. Mariya Moosajee, an ophthalmologist at University College London, used curcumin to prevent cell death in the eyes of tiny zebrafish embryos.1 The experiment showed less cell death and much less severe clefts with curcumin treatment.
The result was especially promising because preventing coloboma in newborns would mean giving treatments to at-risk embryos. ‘India houses the second largest population in the world, and turmeric consumption is widespread even during pregnancy,’ Moosajee explains. ‘As no evidence of associated congenital birth defects or toxicity exists, curcumin has the potential to be used prenatally as a preventative medicine in ophthalmology.’
In the meantime, the wide-ranging potential benefits of turmeric – including lowering the risk of heart disease and cancer – have broken through to the mainstream. Coffee shops sell turmeric lattes and turmeric products are advertised in newspapers. But are these claims realistic? Some now argue that it’s leaving its characteristic bright orange mark on the scientific literature. The colour of turmeric and curcumin are among many problematic properties, potentially causing false positives in drug screening tests that measure fluorescence.
Many other laboratory studies conclude that there are benefits, including Moosajee’s. Curcumin has therefore made its way into clinical trials for many problems ranging from irritable bowel syndrome to prostate cancer. The European Medicines Agency’s Committee on Herbal Medicinal Products (CHMP) allows the use of Javanese turmeric for ‘digestive problems such as feelings of fullness, slow digestion and flatulence’. However with data ‘not sufficient to draw firm conclusions’ the CHMP decision is based on such medicines’ ‘long-standing use’. And while some researchers cite the many clinical trials done with curcumin, it’s also common to hear that even more are needed.
Michael Walters, a medicinal chemist from the University of Minnesota in the US, is therefore sceptical. He asserts that curcumin medicines have ‘never been shown to be conclusively effective in a randomised, placebo-controlled clinical trial’.2 Worse still, a leading advocate of turmeric and curcumin treatments retired while reportedly being investigated by his employer for alleged data manipulation. So can we scrub away the confusion and see what truth lies beneath?
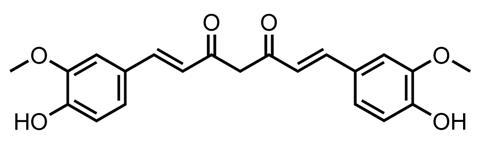
When first using curcumin in experiments Walters found it ‘very difficult to work with’, he tells Chemistry World. In 2014, together with Jonathan Baell from Monash University in Melbourne, Australia, Walters wrote a comment article in Nature about ‘pan-assay interference compounds, or Pains’.3 These are drug lead molecules that succeed disproportionately often – and wrongly – in screening experiments. Curcumin is among the ‘compounds that should ring the most warning bells’, Baell and Walters wrote. Walters says that he therefore asked himself ‘How does that relate to the activity that’s touted so much that it’s commercially available in [health food supermarket] Whole Foods?’
Hyperactive healer
Walters’ team trawled the 15,000 papers describing curcumin’s biological effects, to which around 50 more are added each week. The scientists found that curcumin shows all the Pains problems, including interfering with fluorescence-based screening. So far curcumin has also racked up reports of over 300 different types of biological activity. That makes it the 10th most promiscuous molecule known to science – with none of that top 10 yet successfully becoming a drug lead.
The fact that curcumin’s molecular structure contains an α,β-unsaturated ketone can explain at least some of those laboratory results. This structure makes curcumin highly reactive and likely to covalently attach to proteins and affect their function. Due to toxicity problems caused by paracetamol covalently attaching to liver proteins, medicinal chemists were once wary about such reactivity. But more recently, they have learned to exploit it, both in drugs and as a way to explore biological function. And that’s how Alexander Adibekian, now at Scripps Research Institute in Florida, US, came to also explore curcumin’s promiscuity.
Curcumin is much more reactive than the other natural products we’re testing
Adibekian’s team had devised a method to profile the proteins in living cells that contain the amino acid cysteine.4 Their approach rapidly and selectively labels the cysteines with an ethynyl benziodoxolone probe. The researchers can then either detect the labelled proteins using liquid chromatography paired with tandem mass spectrometry (LC–MS/MS) or add extra labelling molecules using ‘click chemistry’. First this approach lets them build a list of a cell’s cysteine-containing proteins. Then they can start a separate experiment by adding other molecules to cells that might interact with some cysteines. Again adding probe molecules will detect the proteins they can still bind with. Adibekian and colleagues therefore know the missing ones have previously bound to the molecule they added.
‘We chose curcumin to show how efficient our probe is in profiling targets of cysteine-active small molecules, because we were quite sure we would find a series of interesting targets,’ Adibekian says. His team found curcumin targeted 42 of 2689 possible proteins in the cells they studied. ‘It’s much more reactive than the natural products that we’re testing in other projects.’ He also says that curcumin has many other possible protein targets for covalent binding, reacting through other amino acids like lysine.
Fierce in the lab, feeble in the body
Walters and colleagues say that the lack of selectivity that allows curcumin to bind to so many protein targets makes it ‘pharmacodynamically fierce’. But they write that it is also ‘pharmacokinetically feeble’, partly because its stability is ‘abysmal relative to commercial drugs’. Behind this medicinal chemistry jargon could be the essence of curcumin’s problem.
Pharmacodynamic fierceness is what mainly drives the results people get in the lab. Pharmacokinetic feebleness means curcumin will likely struggle to have effects in people. For example, at room temperature and neutral pH, half of a solution of curcumin will break down in around 20 minutes. It therefore seems unlikely to be in people’s bloodstreams for long. It is more stable in acidic conditions, which is unfortunate, because it’s also much less soluble in that environment. That insolubility could make it tricky for curcumin to pass from our digestive systems into our bloodstreams in the first place.
The curcumin clinical trial found no beneficial effects
Due to curcumin’s pharmacokinetic feebleness, clinical trials can end up using very high doses. Last year, one 600-person placebo-controlled randomised trial in Canadian hospitals sought to exploit curcumin’s reported anti-inflammatory properties during surgery to reduce the chances of post-operation problems.5 The treatment group was given two grams of curcumin eight times over four days around the operation. The trial found no beneficial effects and, perhaps due to the high dose of a reactive substance, a slightly higher risk of short-term kidney injury.
Unusually, the deputy editors of the Canadian Medical Association Journal, where it was published, asked its readers to ‘take turmeric with a grain of salt’. ‘Consumers may still prefer a turmeric to a pumpkin spice latte, but at least they will now be able to hand over their money knowing that a proven health benefit is lacking,’ they concluded.
Some chemists have been trying to resolve the insolubility problem that might prevent turmeric getting into patients’ bloodstreams. Ashwini Nangia from the University of Hyderbad in India is an expert in crystal engineering and has developed soluble versions of curcumin. His research in this area has covered either different crystal polymorphs6 or cocrystals with other substances.7
More trials needed
Nangia’s confidence in turmeric’s medical capabilities is unshaken by the physical and chemical difficulties reported for curcumin, which he calls a ‘wonder biomolecule’. Although his team’s soluble polymorphs and cocrystals of curcumin are not yet sold commercially, he notes that soluble forms are available, including ‘nanocurcumin’. ‘The volume and quality of data needs to be assessed in totality,’ Nangia adds. ‘The benefits of curcumin and turmeric stand out.’
It could be that individual trials like last year’s disappointing one in Canada are misleading. Bharat Aggarwal, formerly of the University of Texas MD Anderson Cancer Center the US, was involved with that trial. He has long been a champion of turmeric’s medicinal role and now runs his own Inflammation Research Center in San Diego, US.
‘There are 350 clinical trials that have been completed with curcumin,’ Aggarwal tells Chemistry World. In one review that Aggarwal co-authored, he argued that they ‘clearly show its safety, tolerability and its effectiveness against various chronic diseases in humans’.8 ‘However, more clinical trials in different populations are necessary to prove its potential against different chronic diseases in humans,’ Aggarwal and colleagues continued.
Aggarwal also argues that curcumin’s ability to hit many biological targets is beneficial. For example, binding to the blood protein albumin could help distribute curcumin through the body and protect it from breakdown, he suggests. Aggarwal argues that albumin could then pass curcumin to other proteins that bind it more strongly.
Yet Aggarwal’s involvement adds an extra scattering of spice to the murky picture in this area. In 2015 he retired from his role at MD Anderson, which was reportedly investigating him for alleged manipulation of research data, typically reusing images to show different results. 28 of the papers he co-authored have been retracted for such reasons, 10 others have received expressions of editorial concern and 17 others have been corrected.
Of his 700 papers listed on the journal database Scopus, 91 contain the word curcumin in the title and are not retraction or errata notices. Six of these have been retracted, three corrected, with one expression of concern. However Aggarwal tells Chemistry World that the problems don’t affect the reproducibility of his findings, and that his retirement wasn’t related to them.
And there may be merit to Aggarwal’s call for more trials. Walters’ team found that from 1995 to 2017 federal US funds exceeding $150 million (£118 million) have been awarded ‘for projects that are linked, directly or indirectly, to the biomedical exploration of curcumin’. Although that sounds a lot, it pales into insignificance compared to mainstream drugs. Typical estimates put the cost of developing a new drug at $2 billion–$3 billion, with the median cost of a single clinical trial being $17 million. By such standards, turmeric research could be considered underfunded, although very few natural products attract this level of spending.
To get the most from existing trials, medical researchers often produce studies known as meta-analyses and systematic reviews. These pool study results to improve their statistical validity. They should consider only trials for one type of medical complaint, rather than trying to provide one single comprehensive curcumin analysis. In these cases less than 20 clinical trials have been run, including at best nearly 1500 patients. Most analyses are tentatively positive, while some are clearly not, and they usually again call for more trials on curcumin.
But there’s a lot of variation in the forms of turmeric and curcumin used, which raises an interesting prospect. What if the idea that only curcumin is behind the golden spice’s health benefits is a red herring? While curcumin is the most abundant compound in turmeric, it only comprises 5% of the total composition of the spice. Even one of the most common scientifically used forms, supplied by laboratory stalwart Sigma-Aldrich, only contains 65% pure curcumin.
Inflammatory findings
That’s a perspective supported by Holger Stark, Annika Frank and colleagues at the University of Düsseldorf in Germany, together with scientists in Palestine and Israel. With herbs and spices commonly used as medicines in the latter countries, Frank explains, they explored how different turmeric extracts affected inflammation mediated through histamine. They found that one made using methanol as a solvent was most active, more so than curcumin itself.9 From that extract, the scientists isolated curcumin and the closely related demethoxycurcumin and bis(4-hydroxycinnamoyl)methane. They all bound similarly tightly to the histamine receptor protein Stark, Frank and colleagues tested them against. The crude extract, however, gave the best results in the binding tests.
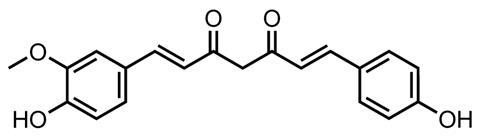
Frank says that some scientists suggest that several components of turmeric work together medicinally. ‘This leads to a challenging task for pharmaceutical industry, as they need to standardise extracts and quantify compounds without knowing which are most relevant,’ she says. Meanwhile, Stark is sceptical about the benefits of turmeric lattes. ‘Such drinks may be delicious but will very unlikely grant health benefits,’ he says. Rather than turn to turmeric, if people are ill he strongly recommends they see their pharmacist or doctor.
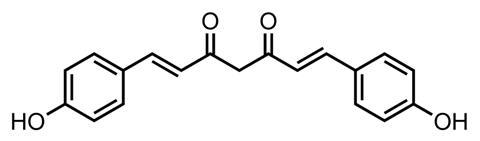
Aggarwal’s team had also earlier published similar findings.10 ‘We have shown that turmeric, curcumin-free turmeric and curcumin, all side-by-side, curcumin is no better than the rest,’ he says. This challenges the pharmaceutical industry differently, Aggarwal adds. ‘If you accept that turmeric can be as good as curcumin, turmeric is $5 a kilo, whereas curcumin is $100 a kilo. So how are people going to make money?’
Moving beyond an exclusive focus on curcumin is a viewpoint where disagreeing scientists like Walters and Aggarwal align. ‘There are 1000 different compounds in turmeric extract, and maybe there’s something in there that is active,’ Walters says. ‘Our point is that reducing turmeric to curcumin might be leading us to miss active components of the extract. Curcumin itself, that structure is probably not the cure for all evil. But there’s probably something in turmeric that may be useful.’
Andy Extance is a science writer based in Exeter, UK
References
1 M Moosajee et al, Hum. Mol. Gen., 2008, 17, 3987 (DOI: 10.1093/hmg/ddn302)
2 K M Nelson et al, J. Med. Chem., 2017, 60, 1620 (DOI: 10.1021/acs.jmedchem.6b00975)
3 J Baell and M Walters, Nature, 2014, 513, 481 (DOI: 10.1038/513481a)
4 D Abegg et al, Angew. Chem. Int. Ed., 2015, 54, 10852 (DOI: 10.1002/anie.201505641)
5 A Garg et al, CMAJ, 2018, 190, E1273 (DOI: 10.1503/cmaj.180510)
6 P Sanphui et al, Chem. Commun., 2011, 47, 5013 (DOI: 10.1039/c1cc10204d)
7 P Sanphui et al, Cryst. Growth Des., 2011, 11, 4135 (DOI: 10.1021/cg200704s)
8 A Kunnumakkara et al, Brit. J. Pharmacol., 2016, 174, 1325 (DOI: 10.1111/bph.13621)
9 A Frank et al, Inflamm. Res., 2017, 66, 923 (DOI: 10.1007/s00011-017-1075-x)
10 B B Aggarwal et al, Mol. Nutr. Food Res., 2013, 57, 1529 (DOI: 10.1002/mnfr.201200838)













2 readers' comments