Theoretical research examining the electric field within the energy-making enzyme ATP synthase alludes to it having a much higher enzymatic efficiency than reported in textbooks.1 Calculations of the enzyme’s molecular electrostatic potential showed changes in the electric field that constructively assist with proton movement alongside ATP formation, showing that the enzyme works over and above its biological catalytic function.
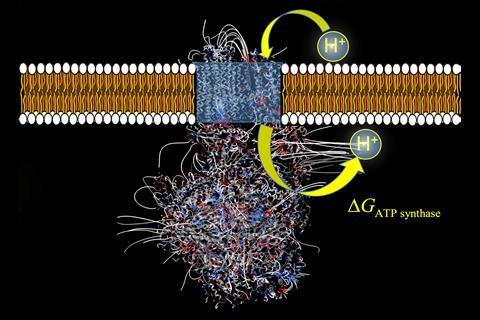
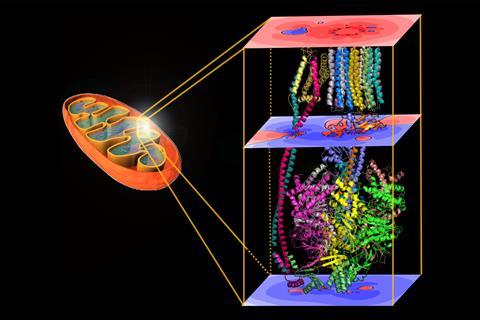
ATP (adenosine triphosphate) is the energy currency in all living cells. The majority of ATP is synthesised in the mitochondria by the enzyme ATP synthase, which completes the last step in a complex electron transfer chain within the inner mitochondrial membrane. To drive the energetically costly conversion of ADP (adenosine diphosphate) and phosphate to ATP, the enzyme makes use of the energy associated with the membrane’s electrochemical gradient created in the electron transfer chain by pumping protons back across the membrane.
Most textbooks quote the efficiency of ATP synthase at around 55%, as calculated from the free energy values of the oxidation of glucose from food to ATP. However, these calculations often don’t consider the role of ATP synthase in transporting protons back across the membrane. To recognise and select protons from the background heat noise for transportation across the membrane, the enzyme must dissipate energy to counteract the decrease in entropy from this proton migration. Now, calculations by Chérif Matta of Mount Saint Vincent University, Canada, and colleagues to determine the electric field within ATP synthase point to an explanation for how ATP synthase functions whilst dissipating this proton selection energy. The calculations also point to a much higher thermodynamic efficiency for ATP synthase.
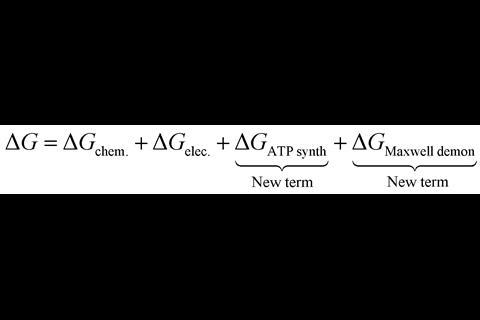
Matta’s team calculated the molecular electrostatic potential of ATP synthase from five different species by calculating the electric field along the direction of proton movement through the enzyme. These calculations showed voltage differences between the points of proton entry and exit through the enzyme and a potential spike at the point of proton entry. Both observations were translated to new free energy terms to add to the equation for the free energy of ATP synthesis.
‘As catalysts, enzymes don’t interfere in the free energy of the reaction, but this is not the case for ATP synthase,’ says Matta. The new free energy terms proposed by Matta’s team correlate with two new roles that can be assigned to ATP synthase beyond catalysing the formation of ATP. Firstly, the potential difference between proton entry and exit adds constructively to the electrochemical gradient of the inner mitochondrial membrane, compensating for the required dissipation of the proton selection energy and altering the free energy of this proton translocation. Secondly, the potential spike at proton entry represents a kinetic barrier, showing ATP synthase as a regulator of proton migration as well. These new roles for the enzyme reinforce earlier estimations that the true efficiency of ATP synthase is around 90%.
Sunil Nath, a senior researcher at the Indian Institute of Technology Delhi who proposed a torsional mechanism for ATP synthesis where the enzyme also makes use of energy from rotatory movement associated with ion translocation,2 notes that ‘an interesting aspect of this work is that the molecular electrostatic potential creates a kinetic barrier for proton translocation through ATP synthase.’ Nath adds that mechanistic studies relating to how this kinetic barrier affects sustained proton migration through the enzyme are necessary to fully understand how energy transduction is achieved by ATP synthase.
Matta is careful to note that these findings are representative for just five ATP synthase structures and emphasises that these electric field calculations are for the enzyme surrounded by water, rather than embedded within the inner mitochondrial membrane. However, the researchers predict larger calculated potential differences when considering the membrane, where the enzyme works even further beyond its catalytic function, so its true enzymatic efficiency could in fact be closer to 100%.
References
1 J-N Vigneau et al, Chem. Commun., 2022, 58, 2650 (DOI: 10.1039/d1cc06793a)
2 S Nath, J. Biol. Phys., 2021, 47, 401 (DOI: 10.1007/s10867-021-09591-8)









No comments yet