No room for the proteins to denature but reactants can still get through
Scientists in Israel have devised a new approach to enzyme immobilisation – encasing them in metallic gold. The caged enzymes are stable at extreme pHs and high temperatures, with one surviving at pH 13.
Immobilised enzymes serve several areas of chemistry, from organic synthesis to biotechnology. The main aims of immobilisation are to stabilise enzymes by preventing their amino acids chains from unfolding, and to make the biocatalysts simple to recover and reuse. Typical immobilisation techniques anchor enzymes to a support. However, it’s inevitable that at least some of the enzymes will detach, and they are still open to attack from the external environment.
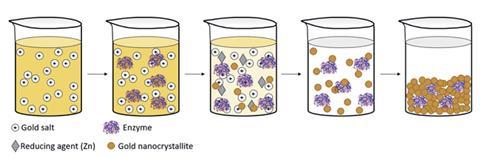
Now, David Avnir and Yael Baruch-Shpigler from the Hebrew University of Jerusalem have shown that metallic gold cages can ensure no part of an enzyme is in contact with the external environment. The research pair made the construct by mixing enzymes with an anionic gold salt followed by a zinc reducing agent to form gold nanocrystallites. These nanocrystallites aggregate into a porous gold cage, which allows reactants to diffuse to and from the enzymes. The gold adheres to the enzymes via surface amino acid residues such as thiols, amines, imines and carboxylate anions.
Avnir and Baruch-Shpigler tested the concept on five industrially important enzymes: glucose oxidase, collagenase, l-asparaginase, horseradish peroxidase and laccase. Gold cages successfully enhanced the thermal stability of all five enzymes. l-Asparaginase could function at pH 13 – conditions that would normally kills the enzyme instantly.
‘Denaturation means that the three-dimensional structure opens and reforms in a different way,’ explains Avnir. ‘The gold cage is so rigid that there is simply no space for the molecule to do the movements that result in denaturation, so that is why it is protected.’
‘Given the chemical inertness of gold metal, such three-dimensional architectures hold great promise to serve as a new type of matrix for enzyme entrapment,’ comments Shengqian Ma, who designs advanced porous materials at the University of South Florida, US.
One of the entrapped enzymes even showed increased activity at higher temperatures, which is a property normally only seen in thermophilic enzymes. ‘It is particularly exciting that at elevated temperatures a collagenase–gold construct shows superior activity than the native enzyme at 25°C,’ notes Imogen Riddell, whose research at the University of Manchester in the UK focuses on building non-natural materials for encapsulating proteins. ‘This highlights the potential for encapsulated enzymes not only as a more selective and environmentally sustainable route to chemical transformations, but also as a more rapid route to synthesis.’
References
This article is open access
Y Baruch-Shpigler and D Avnir, Chem. Sci., 2020, DOI: 10.1039/c9sc05419g









No comments yet